Chapter 11: Q53P (page 589)
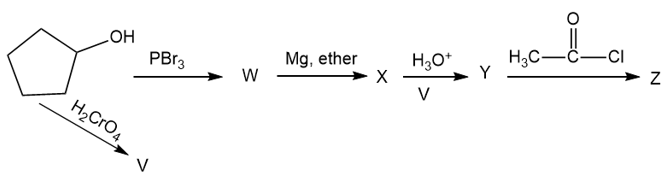
Give the structures of the intermediates and products V through Z.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q53P (page 589)
Give the structures of the intermediates and products V through Z.


All the tools & learning materials you need for study success - in one app.
Get started for free
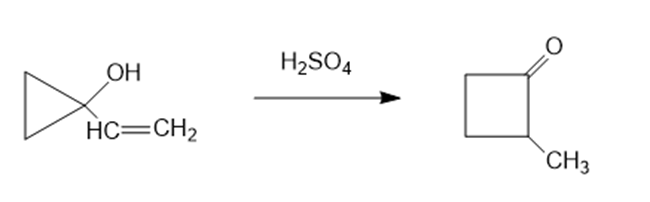
The following reaction involves a starting material with a double bond and a hydroxyl group, yet its mechanism resembles a pinacol rearrangement. Propose a mechanism, and point out the part of your mechanism that resembles a pinacol rearrangement.
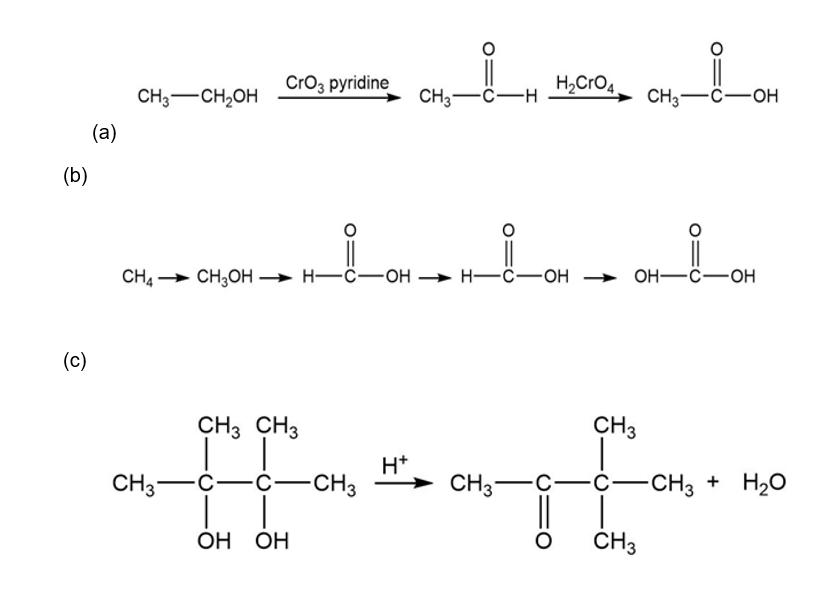
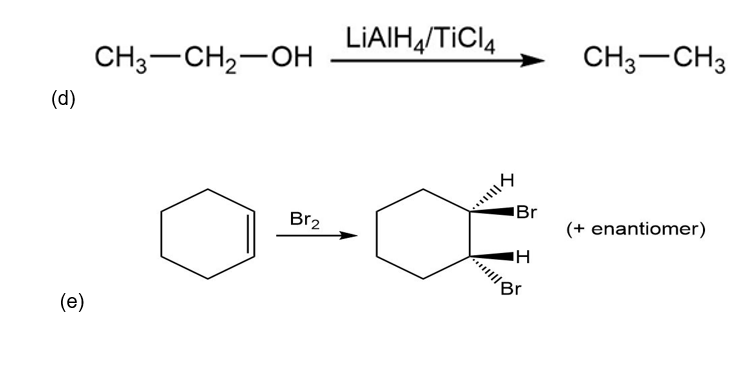
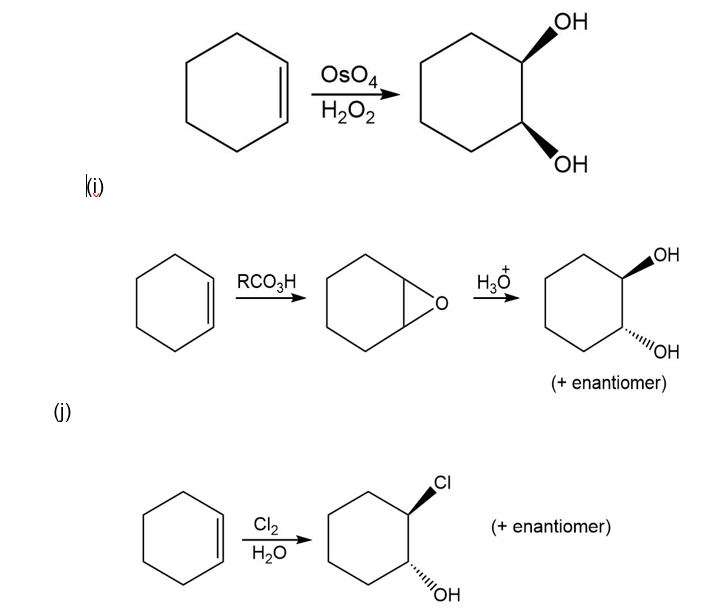
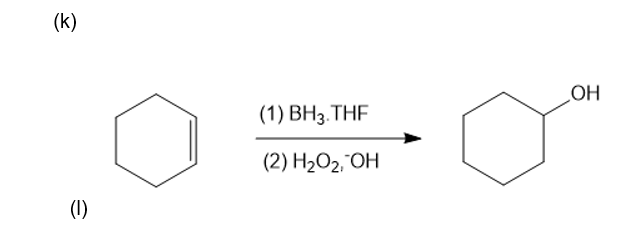
Classify each reaction as an oxidation, a reduction, or neither.
To practice working through the early parts of a multistep synthesis, devise syntheses of
(a) pentan-3-one from alcohols containing no more than three carbon atoms.
(b) 3-ethylpentan-2-one from compounds containing no more than three carbon atoms.
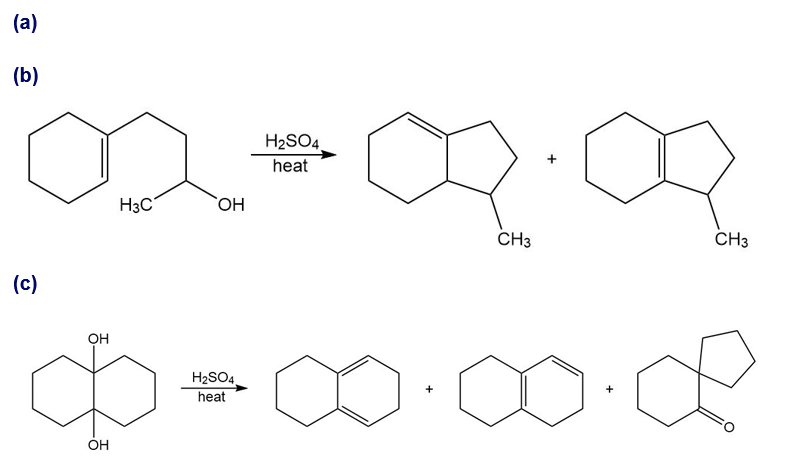
Propose mechanisms for the following reactions. In most cases, more products are formed than are shown here. You only need to explain the formation of the products shown, however.
Both cis- and trans-2-methylcyclohexanol undergo dehydration in warm sulfuric acid to give 1-methylcyclohexene as the major alkene product. These alcohols can also be converted to alkenes by tosylation usingand pyridine, followed by elimination using KOC(CH3)3as a strong base. Under these basic conditions, the tosylate of cis-2-methylcyclohexanol eliminates to give mostly 1-methylcyclohexene, but the tosylate of trans-2-methylcyclohexanol eliminates to give only 3-methylcyclohexene. Explain how this stereochemical difference in reactants controls a regiochemical difference in the products of the basic elimination, but not in the acid-catalyzed elimination.
What do you think about this solution?
We value your feedback to improve our textbook solutions.