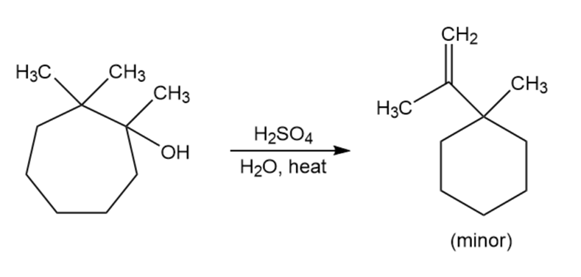
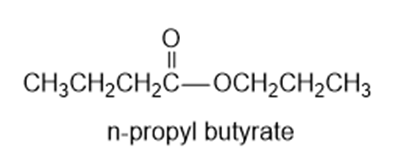
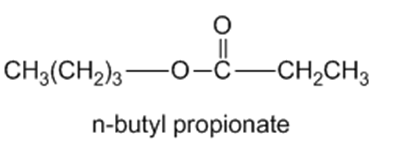
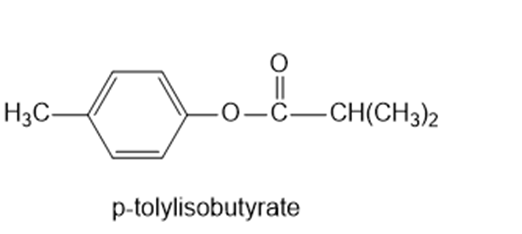
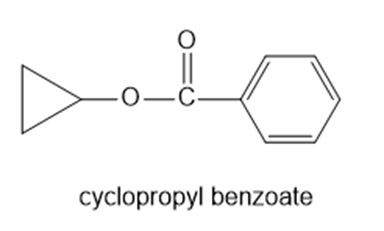
Chapter 11: Q65P (page 591)
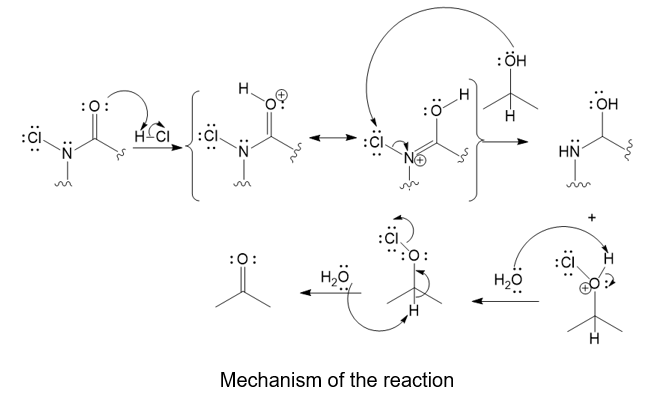
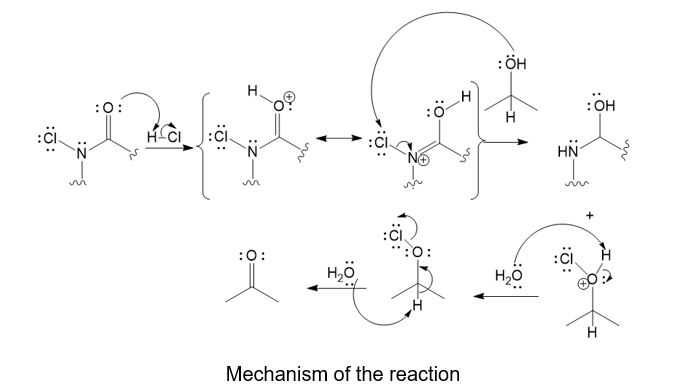
Trichloroisocyanuric acid, TCICA, also known as “swimming pool chlorine,” is a stable solid that oxidizes alcohols, following a mechanism similar to oxidation by HOCl. No reaction occurs between TCICA and the alcohol (in a solvent such as acetonitrile) until one drop of HCl(aq) is added, whereupon the reaction is over in a few minutes. Write the mechanism for this oxidation that shows the key role of the acid catalyst. Show the oxidation of just one alcohol, not three. (Hint: The carbonyls are the most basic sites on TCICA.)
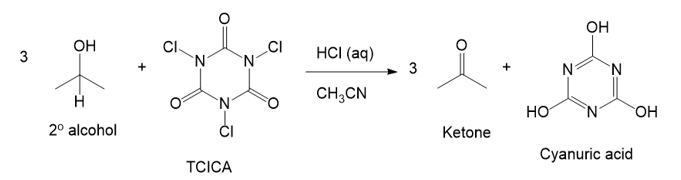
Short Answer