Chapter 11: Q64P (page 591)
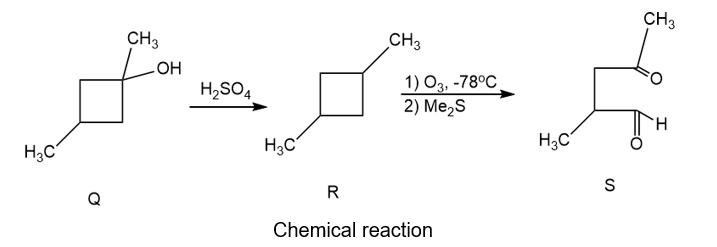
Unknown Q is determined to have a molecular formula of C6H12O. Q is not optically active, and passing it through a chiral column does not separate it into enantiomers. Q does not react with Br2, nor with cold, dilute KMnO4 , nor does it take up H2under catalytic hydrogenation. Heating of Q with H2SO4 gives product R, of formula C6H10, which can be separated into enantiomers. Ozonolysis of a single enantiomer of R produces S, an acyclic, optically active ketoaldehyde of formula C6H10O2. Propose structures for compounds Q, R, and S, and show how your structures would react appropriately to give these results.
Short Answer