Chapter 11: Q46P (page 588)
Show how you would convert (S)-heptan-2-ol to
(a) (S)-2-chloroheptane.
(b) (R)-2-bromoheptane.
(c) (R)-heptan-2-ol.
Short Answer
(a)

(b)
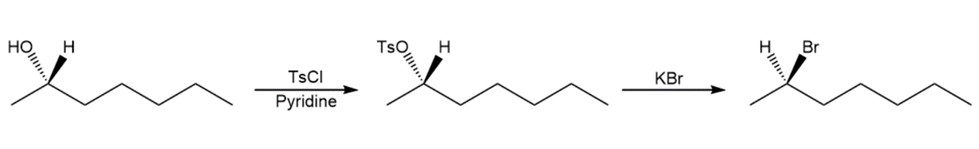
(c)
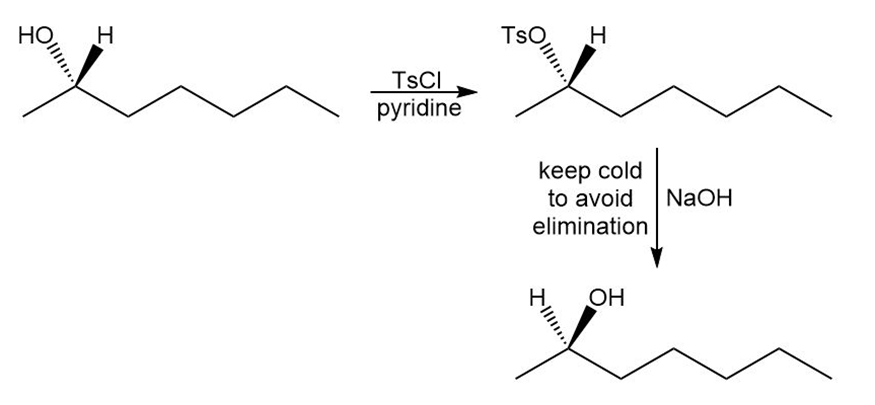
Learning Materials
Features
Discover
Chapter 11: Q46P (page 588)
Show how you would convert (S)-heptan-2-ol to
(a) (S)-2-chloroheptane.
(b) (R)-2-bromoheptane.
(c) (R)-heptan-2-ol.
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use a simple chemical test to distinguish between the following pairs of compounds. Tell what you would observe with each compound.
(a)isopropyl alcohol andtert-butyl alcohol
(b)isopropyl alcohol and butan-2-one, CH3COCH2CH3
(c)hexan-1-ol and cyclohexanol
(d)allyl alcohol and propan-1-ol
(e)butan-2-one andtert-butyl alcohol
Predict the products of the reactions of the following compounds with:
(1) chromic acid or excess sodium hypochlorite with acetic acid.
(2)PCC or NaOCl (1 equivalent) with TEMPO.
(a)cyclohexanol (b)1-methylcyclohexanol
(c)cyclopentylmethanol (d)cyclohexanone
(e)cyclohexane (f)1-phenylpropan-1-ol
(g)hexan-1-ol (h)acetaldehyde, CH3CHO
Contrast the mechanism of the two preceding reactions, the dehydration and condensation of ethanol.
Alcohols combine with ketones and aldehydes to form interesting derivatives, which we will discuss in Chapter 18. The following reactions show the hydrolysis of two such derivatives. Propose mechanisms for these reactions.
(a)
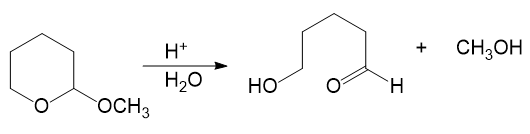
(b)

Give the structure of the principal product(s) when each of the following alcohols reacts with (1) Na2Cr2O7/H2SO4 (2) PCC, (3) DMP, and (4) 1 equiv NaOCl-TEMPO.
(a)octan-1-ol (b)octan-3-ol
(c) 4-hydroxydecanal (d) 1-methylcyclohexan-1,4-diol
What do you think about this solution?
We value your feedback to improve our textbook solutions.