(a)The reaction of cyclohexanol with PCC or NaOCl, TEMPO and H2CrO4 or NaOCl/HOAc produces cyclohexanone as the product. The reaction can be given as:
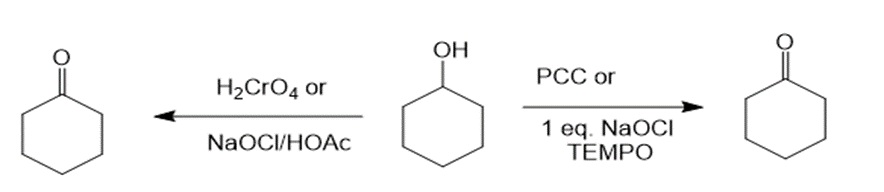
Reaction of cyclohexanol with various reagents
(b)The reaction of 1-methyl cyclohexanol with PCC or NaOCl, TEMPO and H2CrO4 or NaOCl/HOAc creates no products. The reaction can be given as:
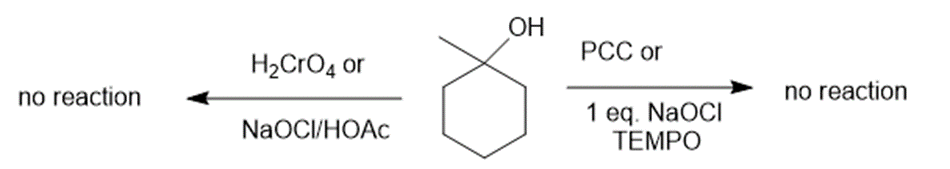
Reaction of 1-methylcyclohexanol with various reagents
(c)The reaction of cyclopentylmethanol with PCC or NaOCl, TEMPO produces cyclopentanal as the product. Cyclopentylmethanol reacts with H2CrO4 or NaOCl/HOAc creating cyclopentanoic acid as the product. The reaction can be given as:

Reaction of cyclopentylmethanol with various reagents
(d) The reaction of cyclohexanone with PCC or NaOCl, TEMPO and H2CrO4 or NaOCl/HOAc creates no products. The reaction can be given as:
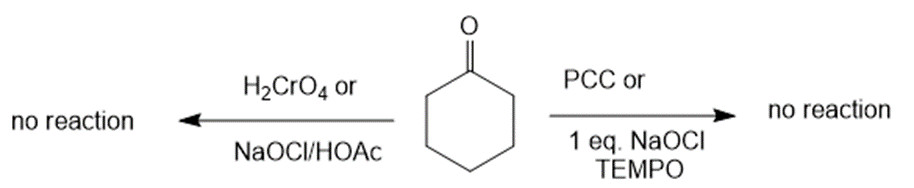
Reaction of cyclohexanone with various reagents
(e) The reaction of cyclohexane with PCC or NaOCl, TEMPO and H2CrO4 or NaOCl/HOAc creates no products. The reaction can be given as:
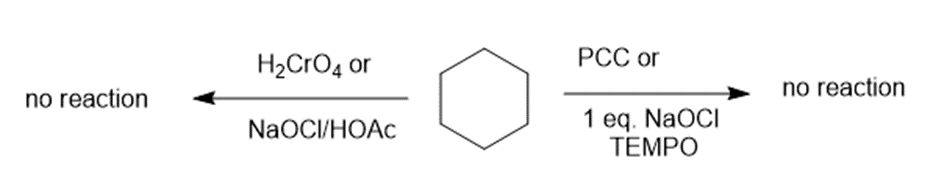
Reaction of cyclohexane with various reagents
(f)1-phenylpropan-1-ol with PCC or NaOCl, TEMPO and H2CrO4 or NaOCl/HOAc generates 1-phenylpropan-1-one as the product.
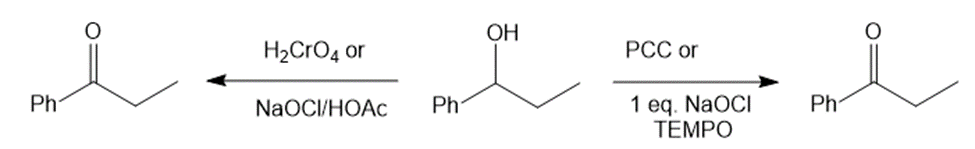
Reaction of 1-phenylpropan-1-ol with various reagents
(g)Hexan-1-ol with PCC or NaOCl, TEMPO generates hexan-1-one as the product. The treatment of Hexan-1-ol with H2CrO4 or NaOCl/HOAc generates hexan-1-oic acid as the product. The reaction can be given as:

Reaction of Hexan-1-ol with various reagents
(h)The reaction of acetaldehyde with PCC produces no products. The reaction of acetaldehyde with NaOCl/TEMPO generates ethanoic acid as the product. Acetaldehyde reacts with H2CrO4 or NaOCl/HOAc generating ethanoic acid as the product. The reaction can be given as:

Reaction of acetaldehyde with various reagents




















