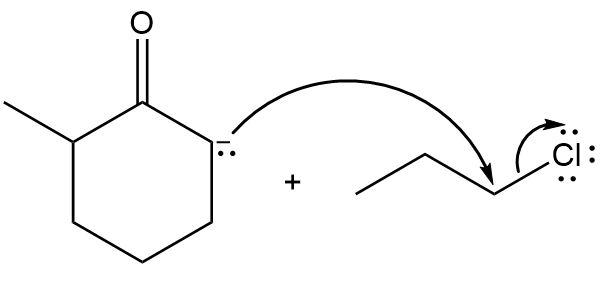
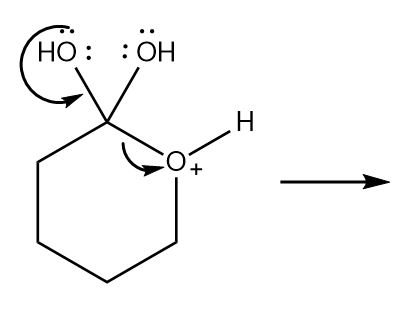
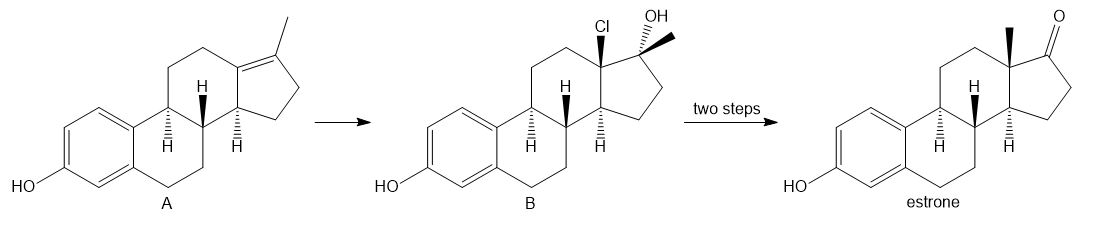
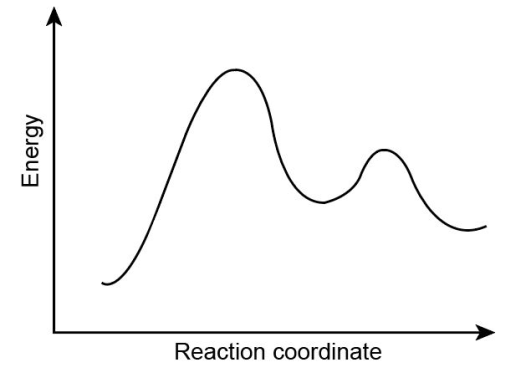
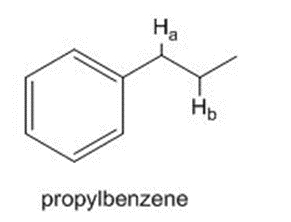
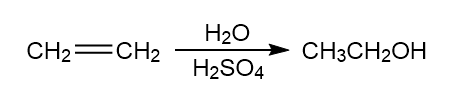
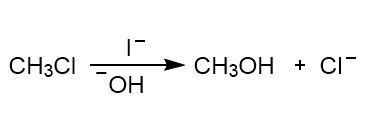
Chapter 6: Problem 6.9 (page 226)
a. Which corresponds to a negative value of localid="1648198317845" , localid="1648198334107" or Keq= 0.001 ?
b. Which corresponds to a lower value oflocalid="1648198364090" , localid="1648198396853" or localid="1648198411007" ?
Short Answer
Answer
a. corresponds to a negative value of .
b. corresponds to a lower value of .