Chapter 6: Problem 6.7 (page 224)
Use the values in Table 6.2 to calculatefor each reaction. Classify each reaction as endothermic or exothermic.
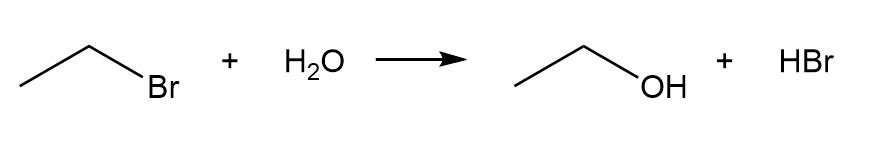
a.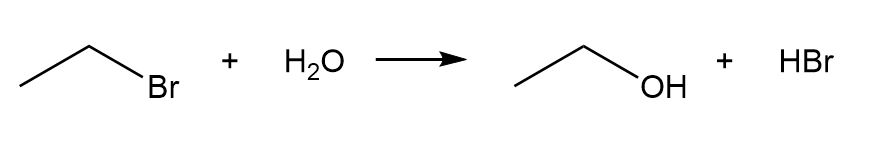
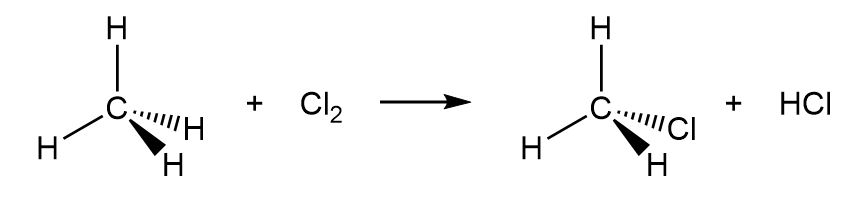
b.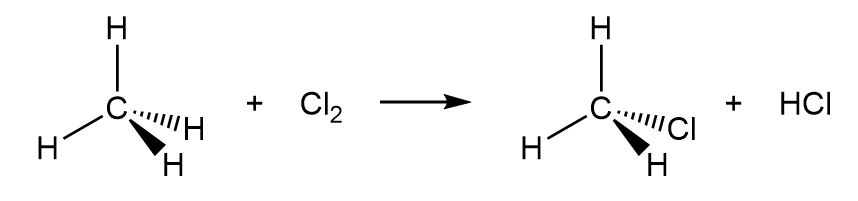
Short Answer
Answer
a. The reaction is endothermic as the of the reaction is positive.
b. The reaction is exothermic as the of the reaction is positive.