Chapter 6: Problem 6.41 (page 243)
For which of the following reactions is a positive value?
a.
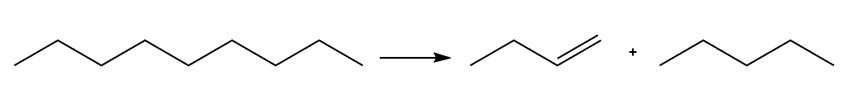
b.
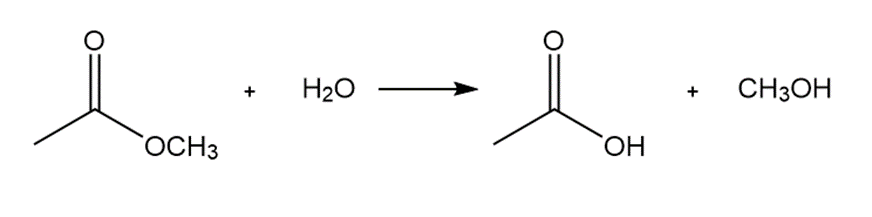
Short Answer
Answer
- is positive.
- No change in .
Learning Materials
Features
Discover
Chapter 6: Problem 6.41 (page 243)
For which of the following reactions is a positive value?
a.
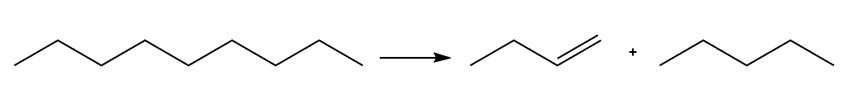
b.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, , and .
a.a concertedwith and .
b. a two-step reaction, , in which the relative energy of the compounds is , and the step is rate-determining.
Calculate Δ��° for each oxidation reaction. Each equation is balanced as written; remember to take into account the coefficients in determining the number of bonds broken or formed.
[ for ; for one C=O in ]
a.role="math" localid="1648191068323"
b.
Draw an energy diagram for the Brønsted–Lowry acid–base reaction of with role="math" localid="1648461988307" to form role="math" localid="1648462071427" and . Label the axes, starting materials, products, Δ��°, and Ea. Draw the structure of the transition state.
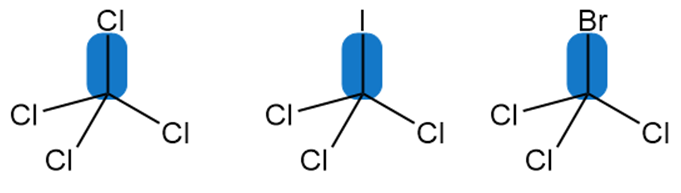
Rank the indicated bonds in order of increasing bond dissociation energy.
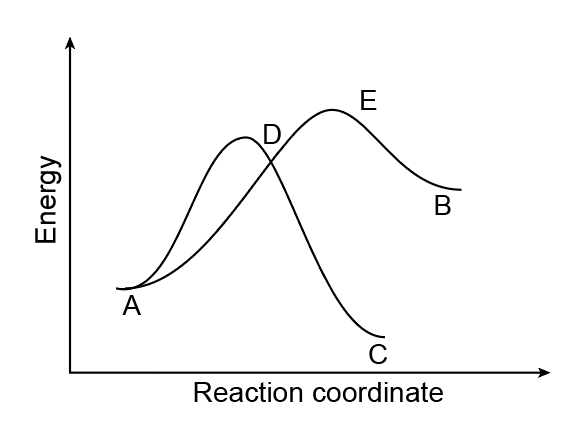
Compound A can be converted to either B or C. The energy diagrams for both processes are drawn on the graph below.
What do you think about this solution?
We value your feedback to improve our textbook solutions.