Chapter 6: Problem 6.34 (page 242)
Rank the indicated bonds in order of increasing bond dissociation energy.
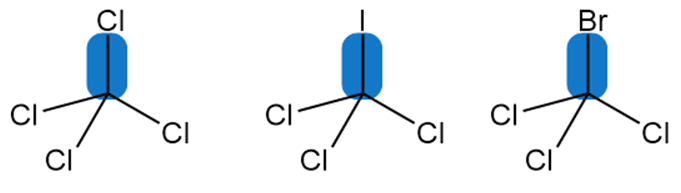
Short Answer
Answer
The bond dissociation energy increases in the order:
Learning Materials
Features
Discover
Chapter 6: Problem 6.34 (page 242)
Rank the indicated bonds in order of increasing bond dissociation energy.

Answer
The bond dissociation energy increases in the order:
All the tools & learning materials you need for study success - in one app.
Get started for free
As we will learn in Section 15.12, many antioxidants—compounds that prevent unwanted radical oxidation reactions from occurring—are phenols, compounds that contain an OH group bonded directly to a benzene ring.
The Diels–Alder reaction, a powerful reaction discussed in Chapter 16, occurs when a 1,3- diene such as A reacts with an alkene such as B to form the six-membered ring in C.
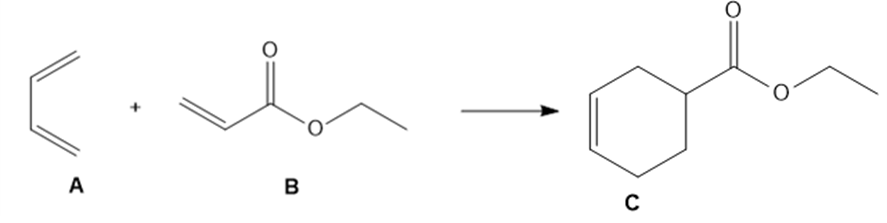
a. Draw curved arrows to show how A and B react to form C.
b. What bonds are broken and formed in this reaction?
c. Would you expect this reaction to be endothermic or exothermic?
d. Does entropy favor the reactants or products?
e. Is the Diels–Alder reaction a substitution, elimination, or addition?
Which bond in each pair has the higher bond dissociation energy?
a.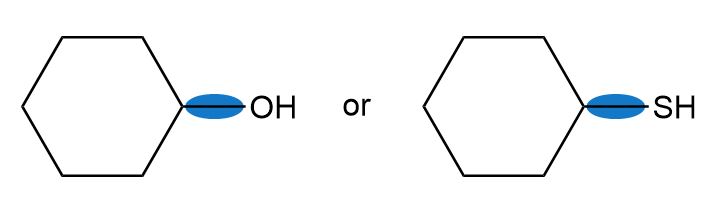
b.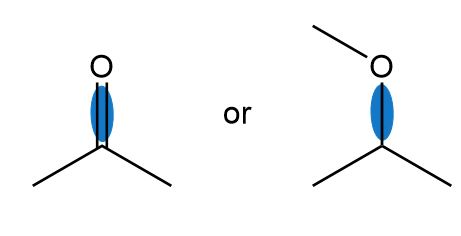
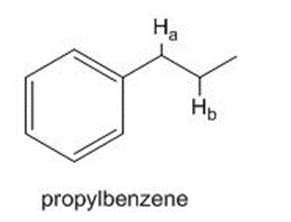
What carbon radical is formed by homolysis of the bond in propylbenzene? Draw all reasonable resonance structures for this radical.
What carbon radical is formed by homolysis of therole="math" localid="1648540916945" bond in propylbenzene? Draw all reasonable resonance structures for this radical.
The bond dissociation energy of one of the C-H bonds is considerably less than the bond dissociation energy of the other. Which C-H bond is weaker? Offer an explanation.
The following is a concerted, bimolecular reaction:.
a. What is the rate equation for this reaction?
b. What happens to the rate of the reaction if is doubled?
c. What happens to the rate of the reaction if [NaCN] is halved?
d. What happens to the rate of the reaction if and [NaCN] are both increased by a factor of five?
What do you think about this solution?
We value your feedback to improve our textbook solutions.