Chapter 6: Problem 6.4 (page 221)
Use curved arrows to show the movement of electrons in each equation.
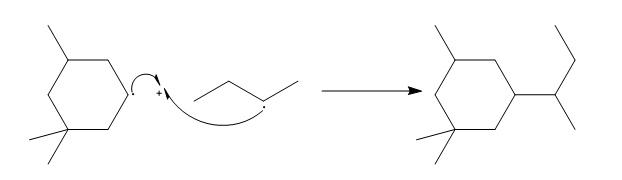
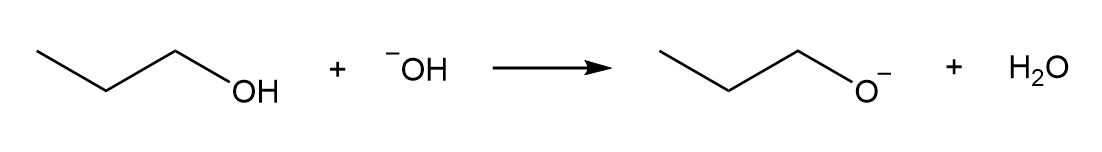
a.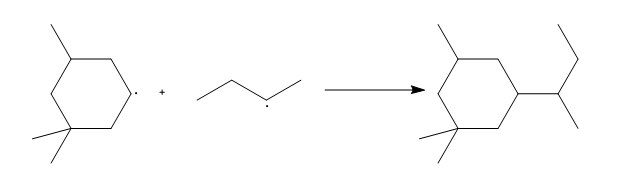
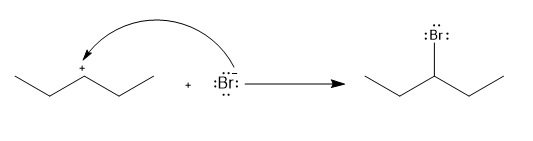
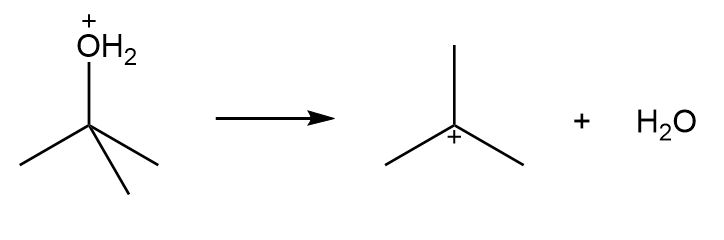
b.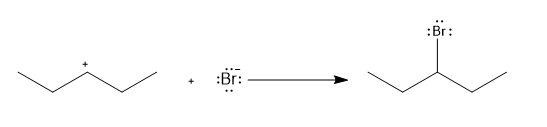
Short Answer
Answer
a.
b.
Learning Materials
Features
Discover
Chapter 6: Problem 6.4 (page 221)
Use curved arrows to show the movement of electrons in each equation.
a.
b.
Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
For each rate equation, what effect does the indicated concentration change have on the overall rate of the reaction?
[1]
[2]role="math" localid="1648280223497"
As we learned in Chapter 4, propane has both and hydrogens.
The following is a concerted, bimolecular reaction:.
a. What is the rate equation for this reaction?
b. What happens to the rate of the reaction if is doubled?
c. What happens to the rate of the reaction if [NaCN] is halved?
d. What happens to the rate of the reaction if and [NaCN] are both increased by a factor of five?
Draw the structure for the transition state in each reaction.
a.
b.
As we will learn in Section 15.12, many antioxidants—compounds that prevent unwanted radical oxidation reactions from occurring—are phenols, compounds that contain an OH group bonded directly to a benzene ring.
What do you think about this solution?
We value your feedback to improve our textbook solutions.