Chapter 6: Problem 6.24 (page 236)
Write a rate equation for each reaction, given the indicated mechanism.
a.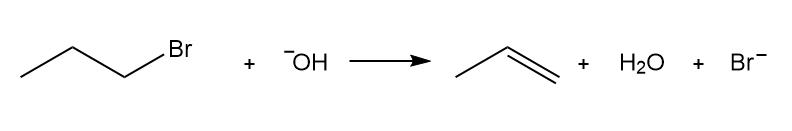
b.
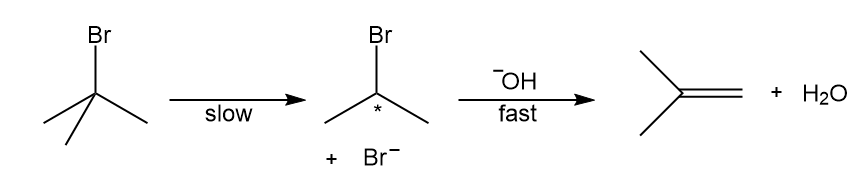
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 6: Problem 6.24 (page 236)
Write a rate equation for each reaction, given the indicated mechanism.
a.
b.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw an energy diagram for the Brønsted–Lowry acid–base reaction of with role="math" localid="1648461988307" to form role="math" localid="1648462071427" and . Label the axes, starting materials, products, ΔH°, and Ea. Draw the structure of the transition state.
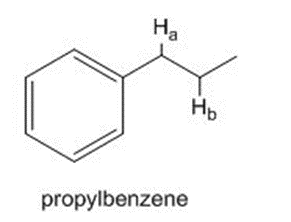
What carbon radical is formed by homolysis of the bond in propylbenzene? Draw all reasonable resonance structures for this radical.
What carbon radical is formed by homolysis of therole="math" localid="1648540916945" bond in propylbenzene? Draw all reasonable resonance structures for this radical.
The bond dissociation energy of one of the C-H bonds is considerably less than the bond dissociation energy of the other. Which C-H bond is weaker? Offer an explanation.
Explain why is more acidic than , even though the C-H bond in has a higher bond dissociation energy than the C-H bond in .
Given each of the following values, is the starting material or product favored at equilibrium?
a.
b.
The equilibrium constant for the conversion of the axial to the equatorial conformation of methoxycyclohexane is 2.7.
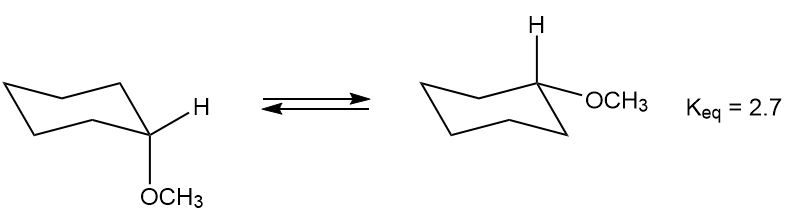
What do you think about this solution?
We value your feedback to improve our textbook solutions.