Chapter 23: Q25. (page 924)
Question:What ketones are prepared by the following reactions?
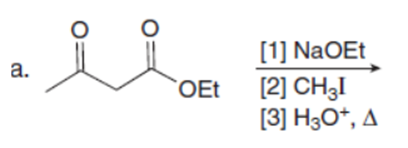
Short Answer
Answers
The ketones prepared from the given reactions are shown below:-
Learning Materials
Features
Discover
Chapter 23: Q25. (page 924)
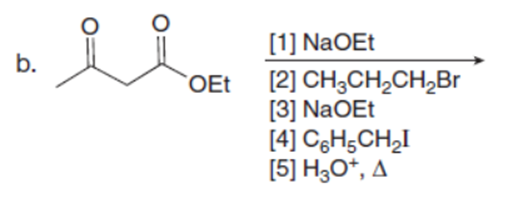
Question:What ketones are prepared by the following reactions?


Answers
The ketones prepared from the given reactions are shown below:-
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (–)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomer isolated from the tailflower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.
Question: Draw the products of each reaction.
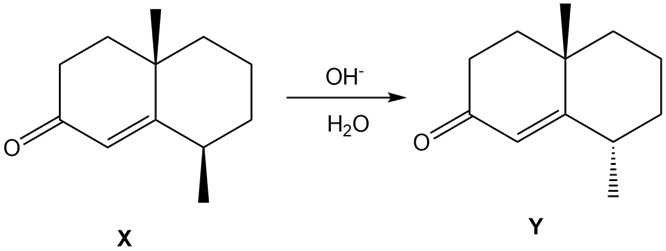
Question: Treatment of α, β-unsaturated carbonyl compound X with base forms the diastereomer Y. Write a stepwise mechanism for this reaction. Explain why one stereogenic center changes configuration but the other does not.
Question: Acid-catalyzed bromination of pentan-2-one (CH3COCH2CH2CH2) forms two products: BrCH2COCH2CH2CH3 (A) and CH3COCH2(Br)CH2CH3(B). Explain why the major product is B, with the Br atom on the more substituted side of the carbonyl group.
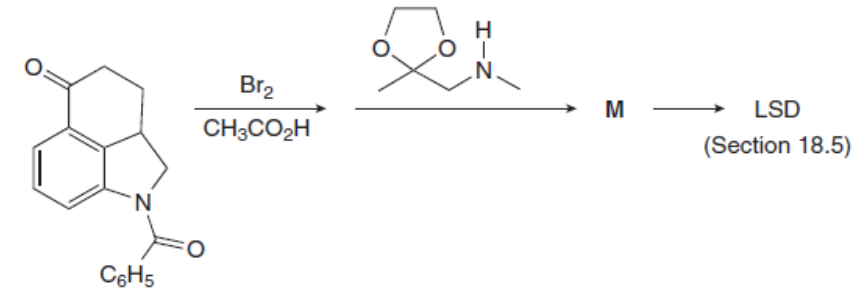
Question: Identify the product M of the following two-step reaction sequence. M was converted to the hallucinogen LSD (Figure 18.4) in several steps.
What do you think about this solution?
We value your feedback to improve our textbook solutions.