Chapter 23: Q11. (page 924)
Question: Draw the products of each reaction.
Short Answer
Answers
The products formed are shown below.
Learning Materials
Features
Discover
Chapter 23: Q11. (page 924)
Question: Draw the products of each reaction.
Answers
The products formed are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
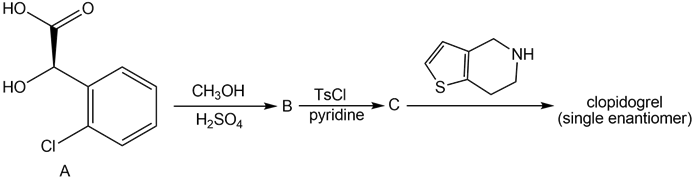
Question: Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S
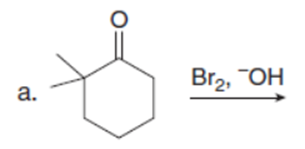
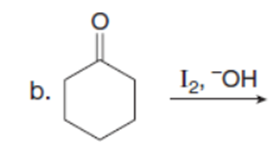
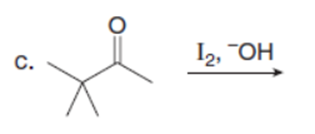
Question: Draw the products of each reaction. Assume excess halogen is present.
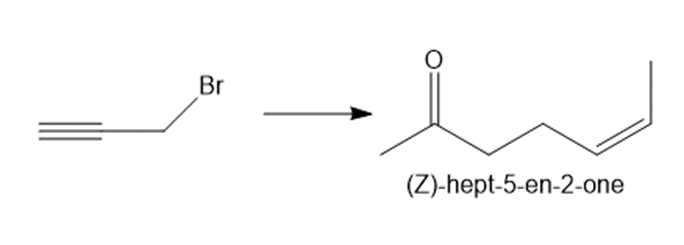
Question: Synthesize (Z)-hept-5-en-2-one from ethyl acetoacetate (CH3COCH2CO2Et ) and the given starting material. You may also use any other organic compounds or required inorganic reagents.
Question: Devise a synthesis of valproic acid [(CH3CH2CH2)2 CHC02H], a medicine used to treat epileptic seizures, using the malonic ester synthesis.
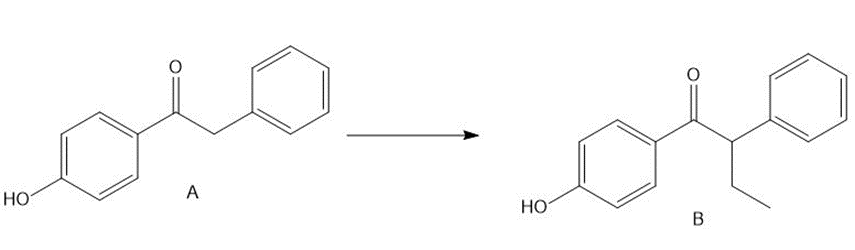
Question: Treatment of ketone A with LDA followed by CH3CH2 did not form the desired alkylation product B. What product was formed instead? Devise a multistep method to convert A to B, a synthetic intermediate used to prepare the anticancer drug tamoxifen (Section 23.8C and the chapter-opening molecule).
What do you think about this solution?
We value your feedback to improve our textbook solutions.