Chapter 1: Q10. (page 20)
Question: Classify each pair of compounds as isomers or resonance structures
a.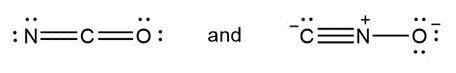
b. 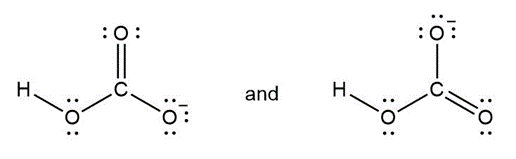
Short Answer
Answer
- Isomers
- Resonance structures
Learning Materials
Features
Discover
Chapter 1: Q10. (page 20)
Question: Classify each pair of compounds as isomers or resonance structures
a.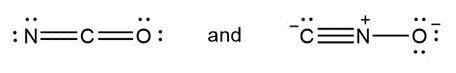
b. 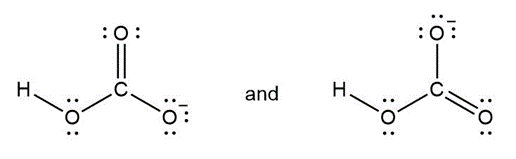
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict all bond angles in each compound.
e.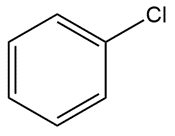
Question: How many covalent bonds are predicted for each atom?
Question: Add curved arrows to show how the first resonance structure can be converted into the second.
a.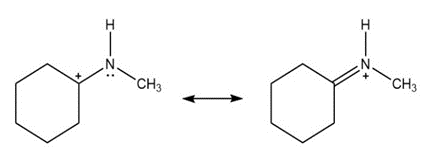
b.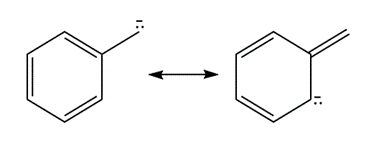
Question: Answer the following questions about acetonitrile :
a. Determine the hybridization of both C and N atoms.
b. Label all bonds as or .
c. In what type of orbital does the lone pair on N reside?
d. Label all bonds as polar or nonpolar.
Question: Rank the following atoms in order of increasing electronegativity. Label the most electronegative and most electropositive atom in each group.
What do you think about this solution?
We value your feedback to improve our textbook solutions.