Chapter 1: Q57. (page 56)
Question: Predict all bond angles in each compound.
e.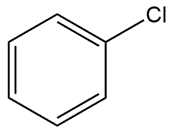
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 1: Q57. (page 56)
Question: Predict all bond angles in each compound.
e.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What orbitals are used to form each bond in the following molecules?
Question: Anacin is an over-the-counter pain reliever that contains aspirin and caffeine. Answer the following questions about each compound:
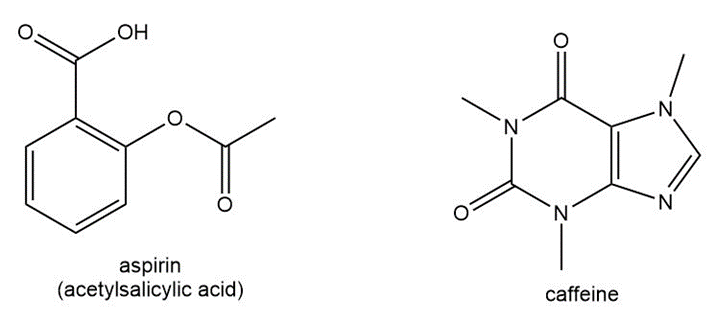
a. What is the molecular formula?
b. How many lone pairs are present on heteroatoms?
c. Label the hybridization state of each carbon.
d. Draw three additional resonance structures.
Question: While the most common isotope of nitrogen has a mass number of 14 (nitrogen-14), a radioactive isotope of nitrogen has a mass number of 13 (nitrogen-13). Nitrogen-13 is used in PET (positron emission tomography) scans by physicians to monitor brain activity and diagnose dementia. For each isotope, give the following information: (a) the number of protons; (b) the number of neutrons; (c) the number of electrons in the neutral atom; (d) the group number; and (e) the number of valence electrons.
Question: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers, or neither.
a. 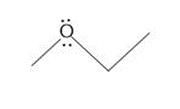
and
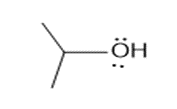
b. and
and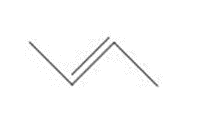
c.  and
and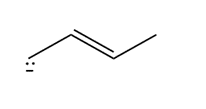
d. and
and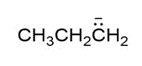
Question: Draw a second resonance structure for each ion. Then, draw the resonance hybrid.
a.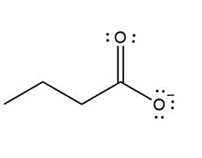
b.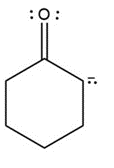
c.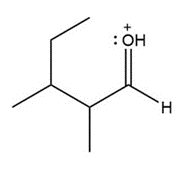
What do you think about this solution?
We value your feedback to improve our textbook solutions.