Chapter 5: Q.49 (page 207)
Draw the structures of (S,S)-ethambutol, a drug used to treat tuberculosis that is 10 times more potant than any of its other isomers.
 ethambutol
ethambutol
Short Answer
Following is the structure of (S,S)-ethambutol:

Learning Materials
Features
Discover
Chapter 5: Q.49 (page 207)
Draw the structures of (S,S)-ethambutol, a drug used to treat tuberculosis that is 10 times more potant than any of its other isomers.
 ethambutol
ethambutol
Following is the structure of (S,S)-ethambutol:

All the tools & learning materials you need for study success - in one app.
Get started for free
Cellulose is water insoluble, despite its many OH groups. Considering its three-dimensional structure, why do you think this is so?
What is the maximum number of stereoisomers possible for each compound?
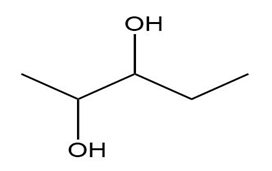
a.

b.
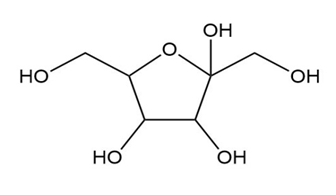
c.
Locate the stereogenic centers in each drug.
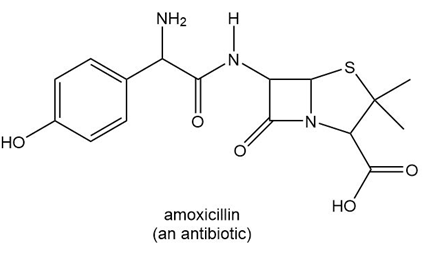
a.
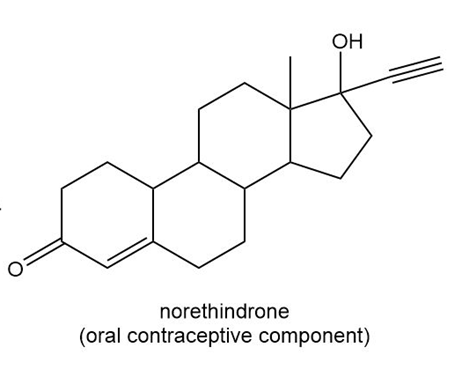
b.

c.
Indicate a plane of symmetry for each molecule that contains one. A molecule may require rotation around a carbon-carbon bond to see the plane of symmetry.
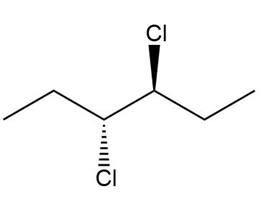
a.

b.

c.
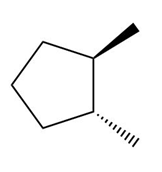
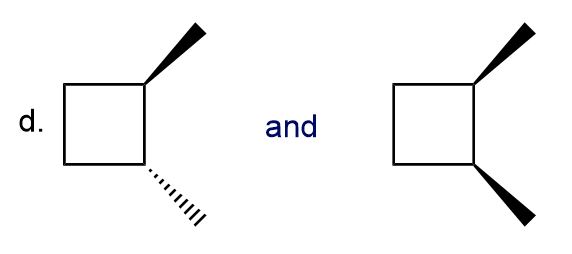
d.
Classify each pair of compounds as constitutional isomers or stereoisomers.
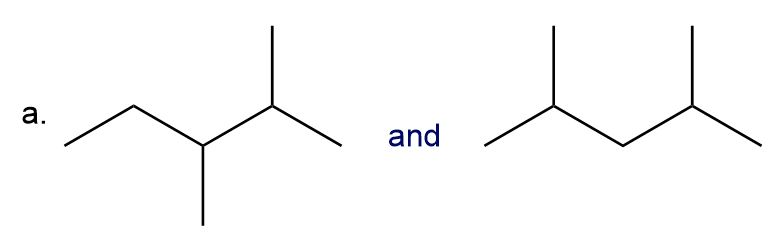
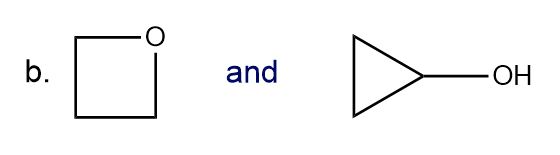

What do you think about this solution?
We value your feedback to improve our textbook solutions.