Chapter 5: Q.13 (page 190)
Label each compound as R or S.
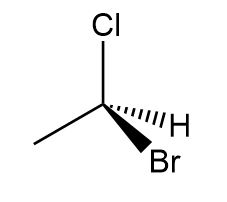
a.
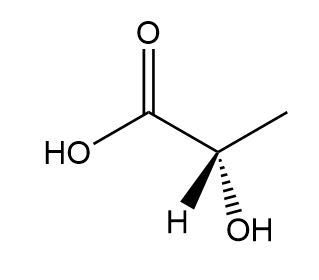
b.
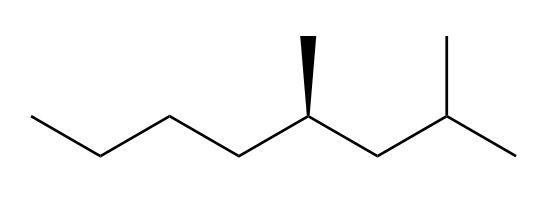
c.
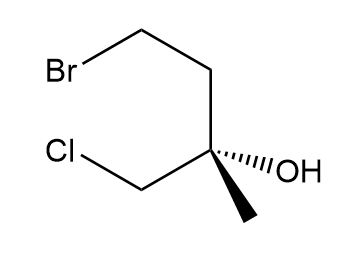
d.
Short Answer
a. S isomer.
b. S isomer.
c. R isomer.
d. S isomer.
Learning Materials
Features
Discover
Chapter 5: Q.13 (page 190)
Label each compound as R or S.

a.

b.

c.

d.
a. S isomer.
b. S isomer.
c. R isomer.
d. S isomer.
All the tools & learning materials you need for study success - in one app.
Get started for free
Cellulose is water insoluble, despite its many OH groups. Considering its three-dimensional structure, why do you think this is so?
Draw all possible constitutional and stereoisomers for a compound of molecular formula having a cyclobutane ring and two methyl groups as substituents. Label each compound as chiral or achiral.
Drawn are four isomeric dimethylcyclopropanes.
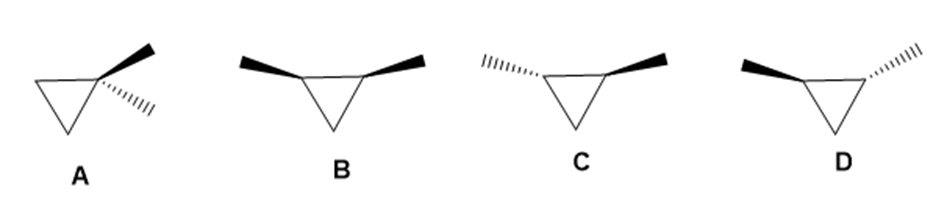
a. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?
b. Label each compound as chiral or achiral.
c. Which compounds, alone, would be optically active?
d. Which compounds have a plane of symmetry?
e. How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D?
f. Which of the compounds are meso compounds?
g. Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?
Determine if each compound is identical to or an enantiomer of A.
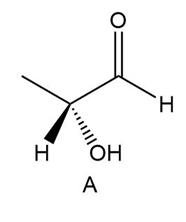
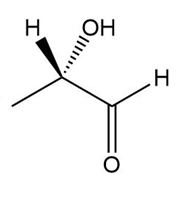
a.
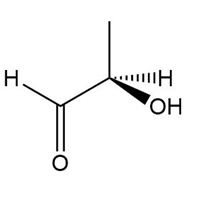
b.
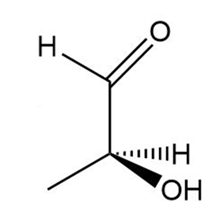
c.
Rank the following groups in order of decreasing priority?
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.