Chapter 18: 18-31P (page 677)
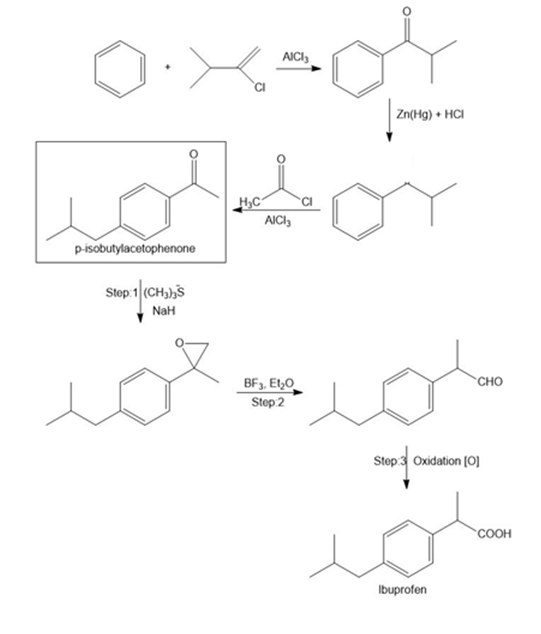
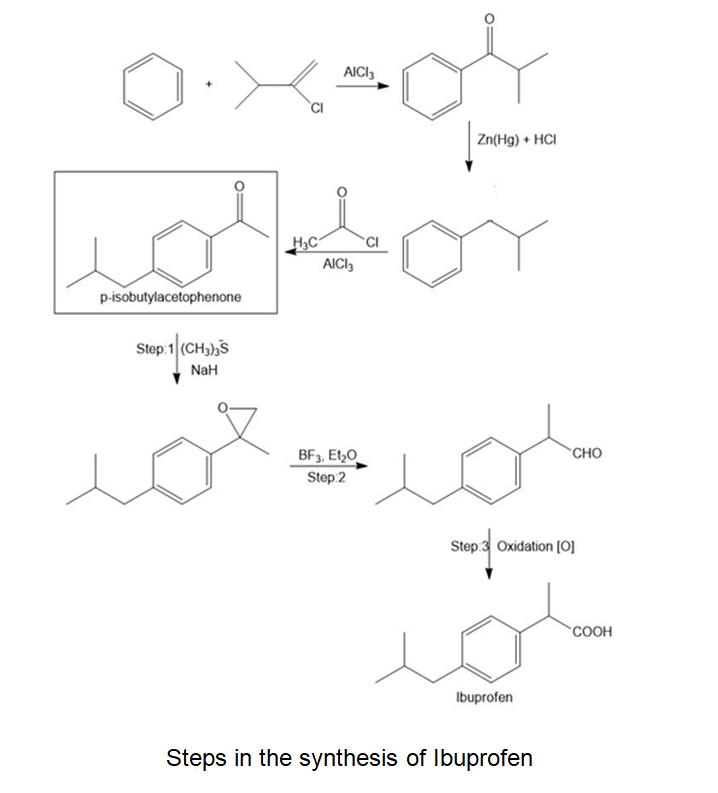
What steps are needed to convert benzene into p-isobutyl acetophenone, a synthetic intermediate used in the synthesis of the anti-inflammatory agent ibuprofen.

Short Answer
Steps in the synthesis of Ibuprofen