Chapter 15: Q8. (page 570)
Question: Which C-H bond in each compound most readily broken during radical halogenation?
a.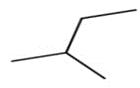
b.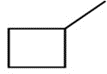
c.
Short Answer
Answer
a. 30C-H bond
b. 30C-H bond
c. 20C-H bond
Learning Materials
Features
Discover
Chapter 15: Q8. (page 570)
Question: Which C-H bond in each compound most readily broken during radical halogenation?
a.
b.
c.
Answer
a. 30C-H bond
b. 30C-H bond
c. 20C-H bond
All the tools & learning materials you need for study success - in one app.
Get started for free
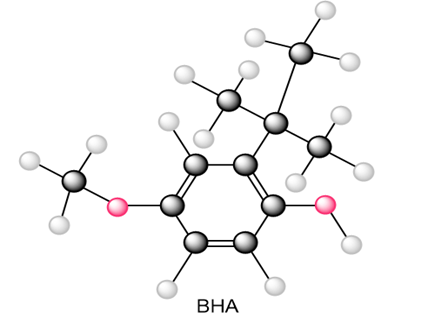
Question: Draw all resonance structures of the radical that results from abstraction of a hydrogen atom from the antioxidant BHA (butylated hydroxy anisole).
Question: As we will learn in Chapter 30, styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic polymerization is an example of electrophilic addition to an alkene involving carbocations.
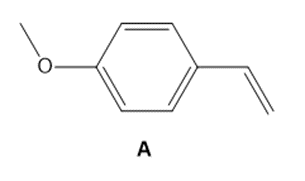
a. Draw a short segment of the polymer formed by the polymerization of A.
b. Why does A react faster than styrene (C6H5CH=CH2)in a cationic polymerization?
Question: When HBr adds to (CH3)2C=CH2 under radical conditions, two radicals are possible products in the first step of chain propagation. Draw the structure of both radicals and indicate which one is formed. Then draw the preferred product from HBr addition under radical conditions.
Question: Devise a synthesis of CH2 CH2CH2CH2Br from HCCH You may use any other required organic or inorganic reagents.
Question: (a) Ignoring stereoisomers, what two allylic hydroperoxides are formed by the oxidation of hex-1-ene with O2? (b) Draw a stepwise mechanism that shows how these hydroperoxides are formed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.