Chapter 12: Q.57. (page 491)
Question: Draw the product of each asymmetric epoxidation reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.57. (page 491)
Question: Draw the product of each asymmetric epoxidation reaction.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
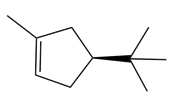
Question: Given that syn addition of H2 occurs from both sides of a trigonal planar double bond, draw all stereoisomers formed when each alkene is treated with H2 .
a.
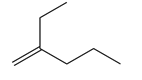
b.
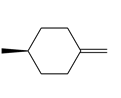
c.
Question: Which alkene in each pair has the larger Heat of hydrogenation?
a.
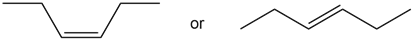
b.
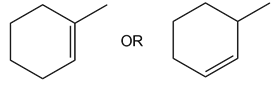
Question: It is sometimes necessary to isomerize a cis alkene to a trans alkene in a synthesis, a process that cannot be accomplished in a single step. Using the reactions that you have learned in Chapters 8–12, devise a stepwise method to convert cis-but-2-ene to trans-but-2-ene.
Question: Which alkyne has the smaller Heat of hydrogenation ? Explain your choice.
Question: Draw the organic products in each of the following reactions.
a.
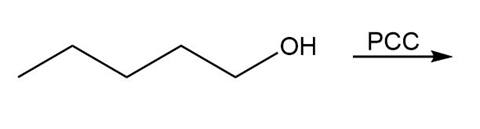
b.
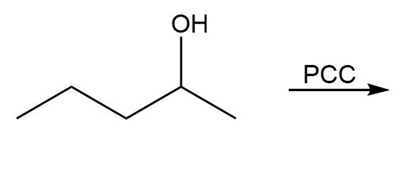
c.
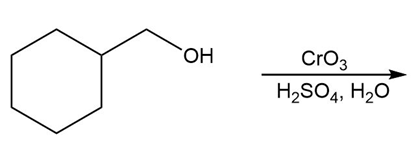
d.
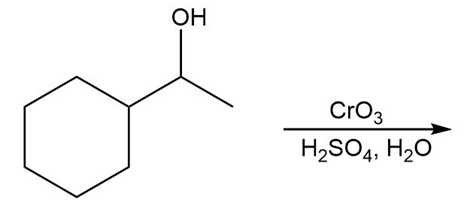
What do you think about this solution?
We value your feedback to improve our textbook solutions.