Chapter 14: Q.21558-14-6P. (page 535)
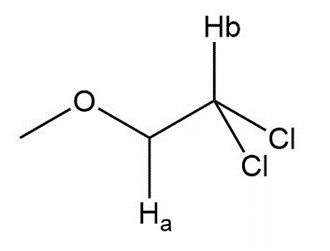
Label the protons in each highlighted CH2 group as enantiotopic, diastereotopic, or homotopic.
a.
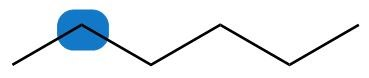
b.
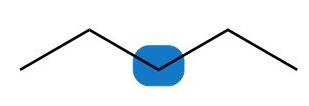
c.
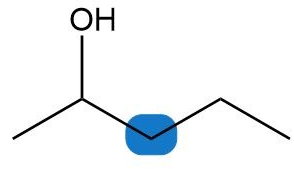
Short Answer
a. Enantiotopic
b. Neither
c. Diastereotopic
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-6P. (page 535)
Label the protons in each highlighted CH2 group as enantiotopic, diastereotopic, or homotopic.
a.

b.

c.

a. Enantiotopic
b. Neither
c. Diastereotopic
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B).
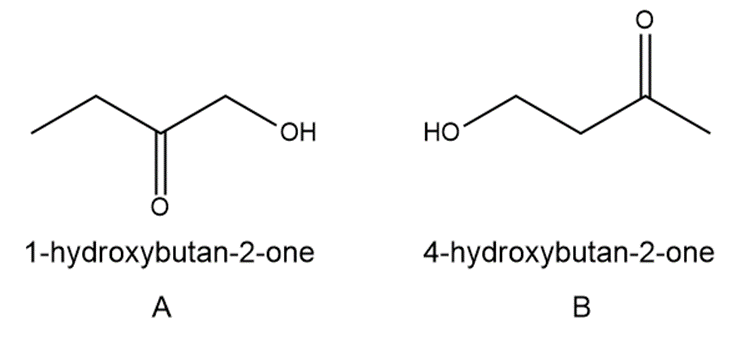
a. What is the molecular ion in the mass spectrum?
b. What IR absorptions are present in the functional group region?
c. How many lines are observed in the 13C NMR spectrum?
d. How many signals are observed in the 1H NMR spectrum?
e. Give the splitting observed for each type of proton as well as its approximate chemical shift.
Question: Identify the structures of isomers E and F (molecular formula C4H802 ). Relative areas are given above each signal.
a.
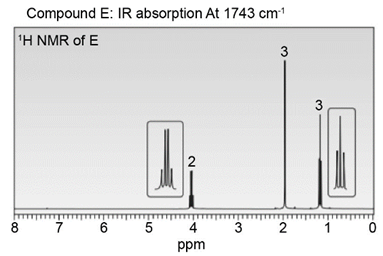
b.
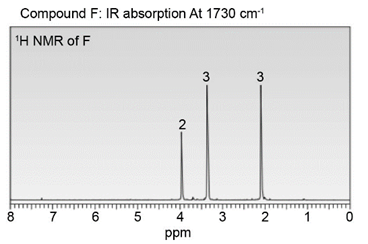
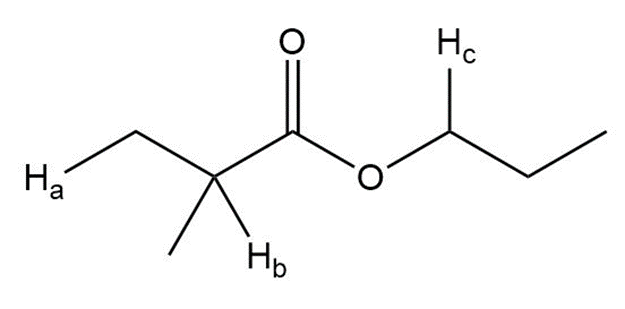
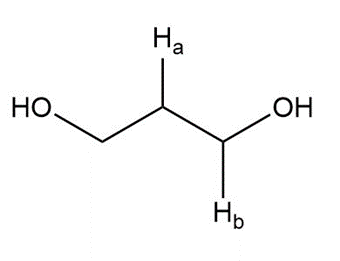
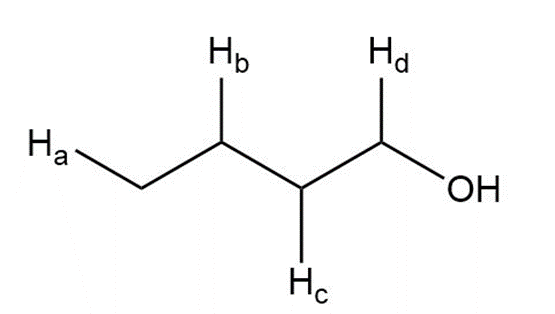
Describe the 1HNMR spectrum of each compound. State how many NMR signals are present, the splitting pattern for each signal, and the approximate chemical shift.
a.
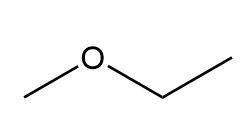
b.
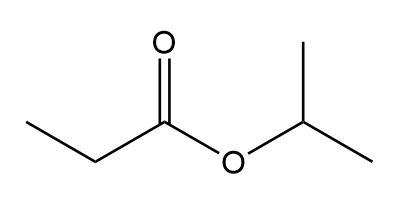
c.
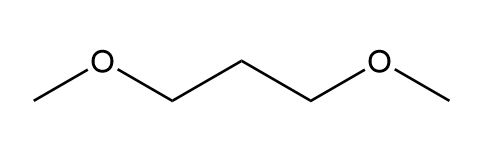
d.
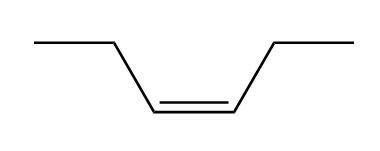
a.
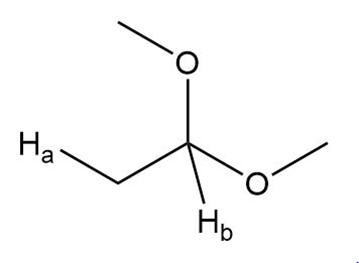
b.
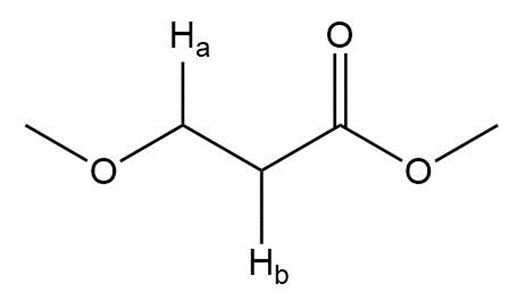
c.
d.
e.
f.
g.
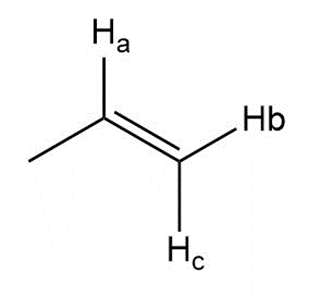
h.
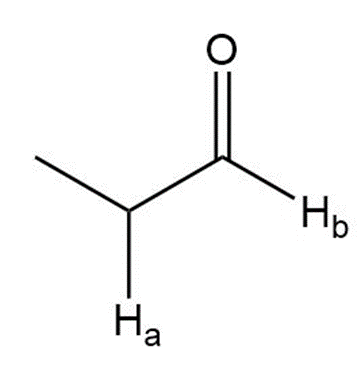
i.
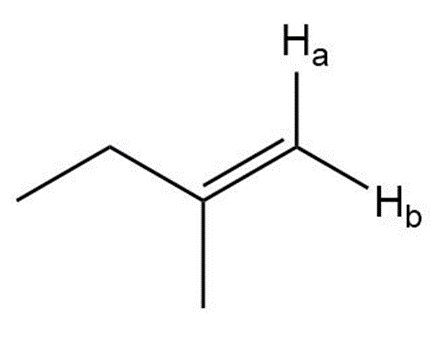
j.
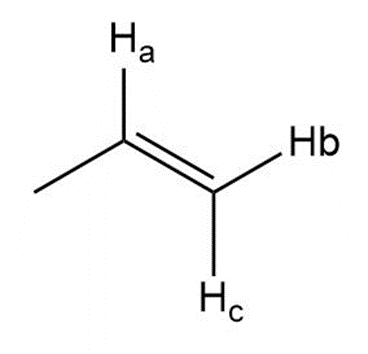
What do you think about this solution?
We value your feedback to improve our textbook solutions.