Chapter 31: Question 31.3 (page 1237)
Draw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B and C in order of increasing melting point.
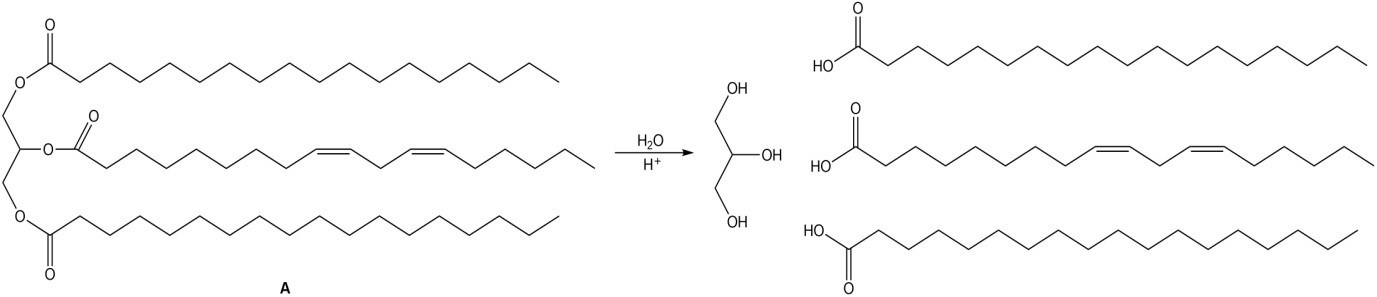
a.,
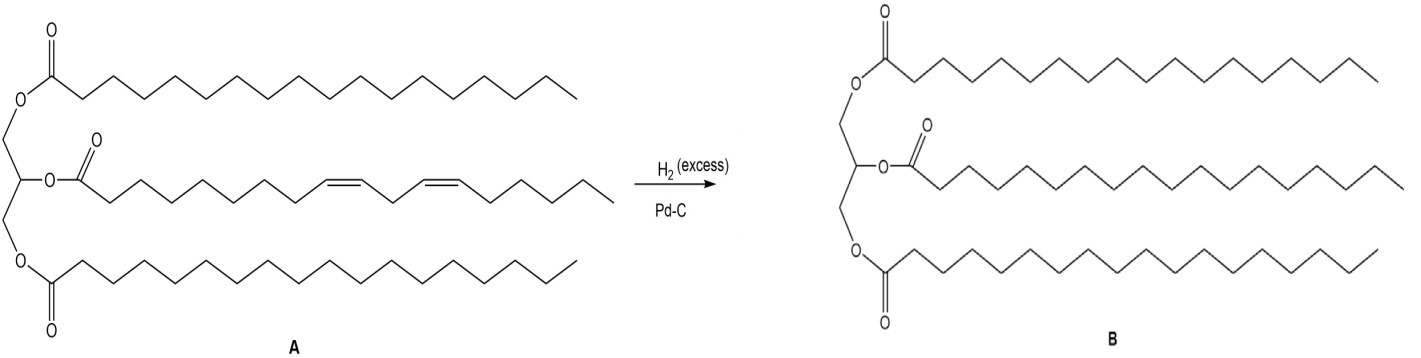
b. (excess),
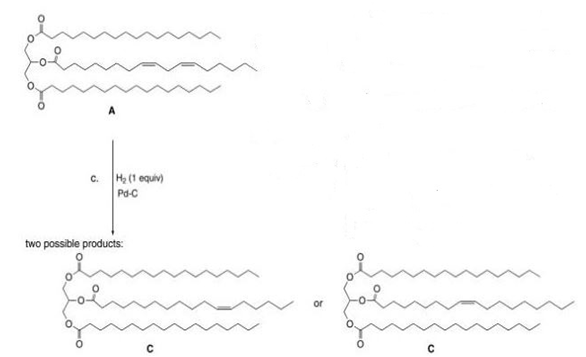
c.(1 equiv),
Short Answer
Answer
a.
b.
c.
Learning Materials
Features
Discover
Chapter 31: Question 31.3 (page 1237)
Draw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B and C in order of increasing melting point.
a.,
b. (excess),
c.(1 equiv),
Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Locate the isoprene units in each compound.
a.
b.
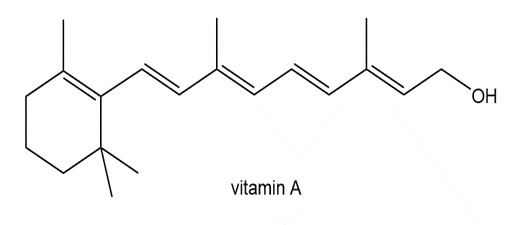
c.
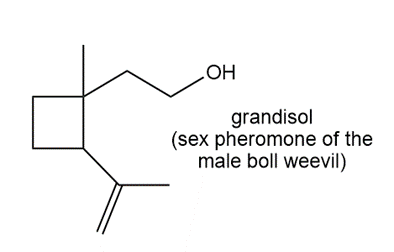
d.
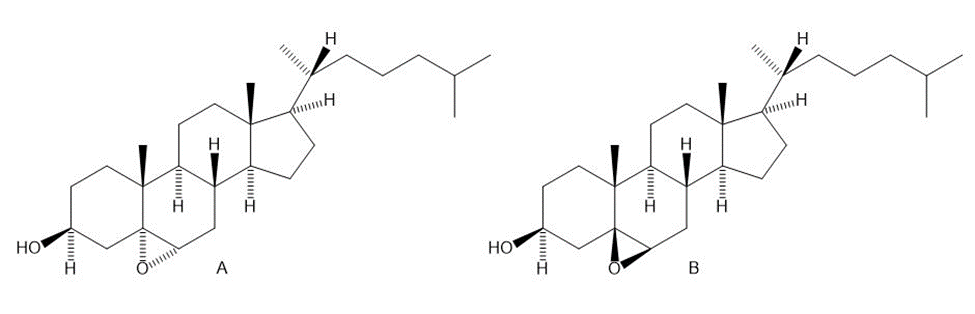
Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the stereochemistry drawn. Why isn’t the isomeric epoxide B formed to any extent?
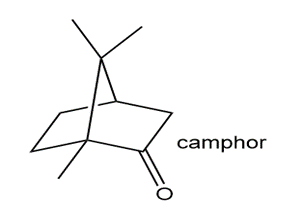
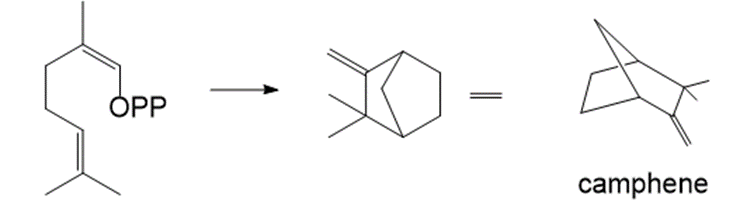
Draw a stepwise mechanism for the following conversion, which forms camphene. Camphene is a component of camphor and citronella oils.
A flask contains 1/3 mole of H2 and 2/3 mole of He. Compare the force on the wall per impact of H2 relative to that for He.
Draw all possible constitutional isomers of a triacylglycerol formed from one mole each of palmitic, oleic, and linoleic acids. Locate the tetrahedral stereogenic centers in each constitutional isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.