Chapter 31: Q 31.10 (page 1246)
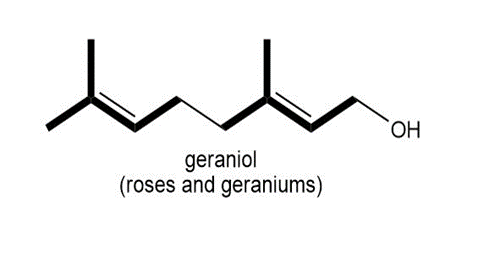
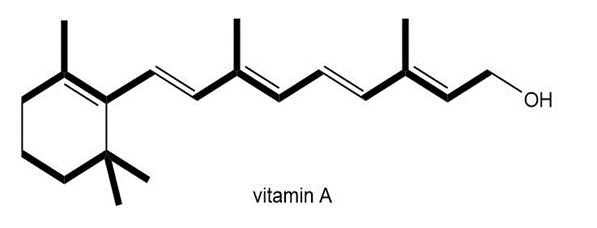
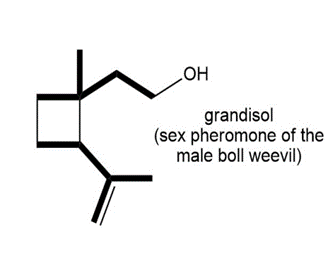
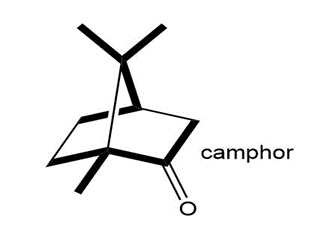
Locate the isoprene units in each compound.
a.
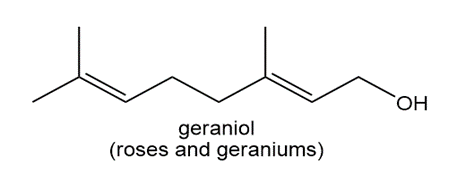
b.
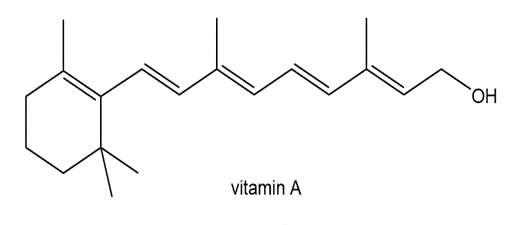
c.
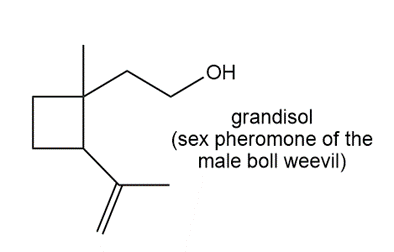
d.
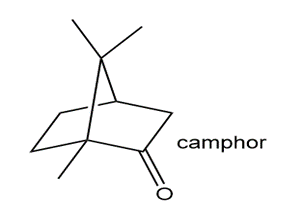
Short Answer
Answer
a.
b.
c.
d.
Learning Materials
Features
Discover
Chapter 31: Q 31.10 (page 1246)
Locate the isoprene units in each compound.
a.

b.

c.

d.

Answer
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
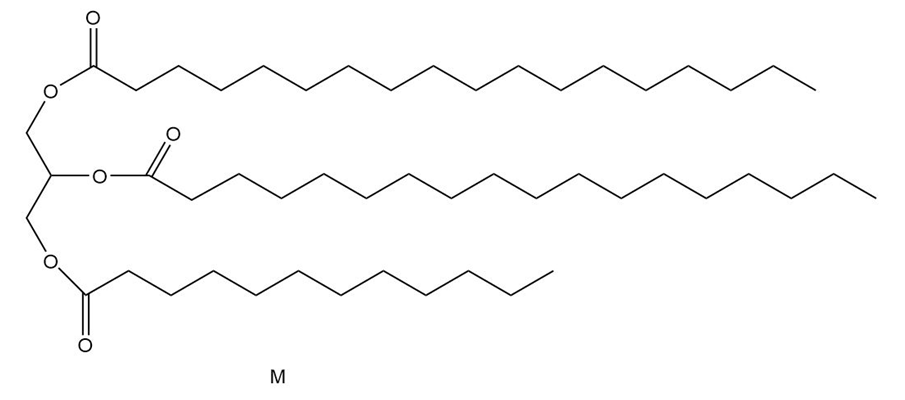
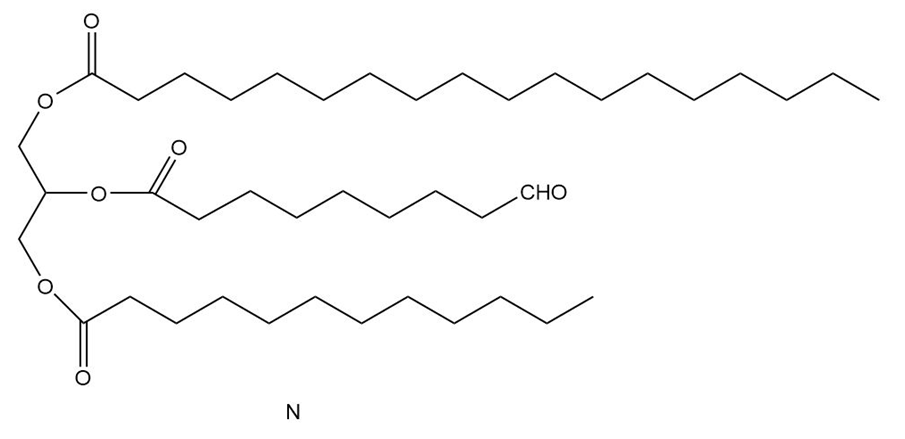
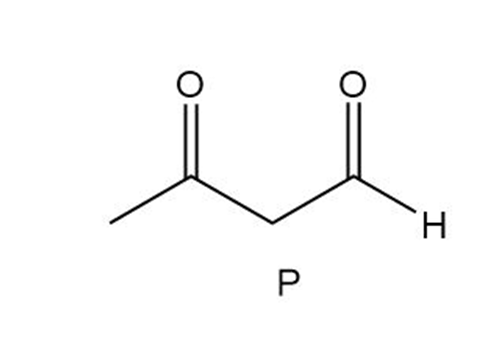
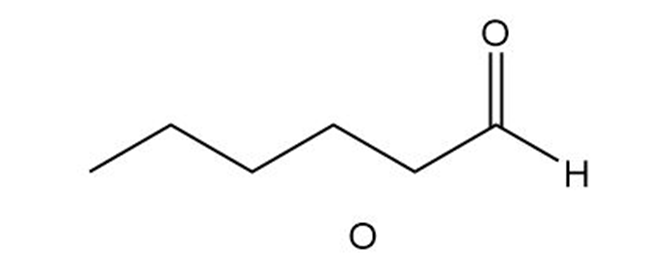
Question:Triacylglycerol L yields compound M when treated with excess , Pd-C. Ozonolysis of L ([1] ; [2] affords compounds N-P. What is the structure of L?
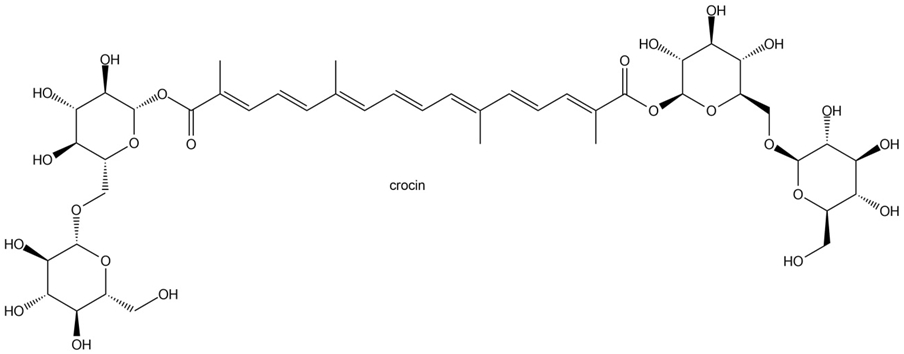
Crocin, which occurs naturally in crocus and gardenia flowers, is primarily responsible for the color of saffron.
(a) What lipid and monosaccharides are formed by the hydrolysis of crocin?
(b) Classify the lipid as a monoterpenoid, diperpenoid, etc., and locate the isoprene units.
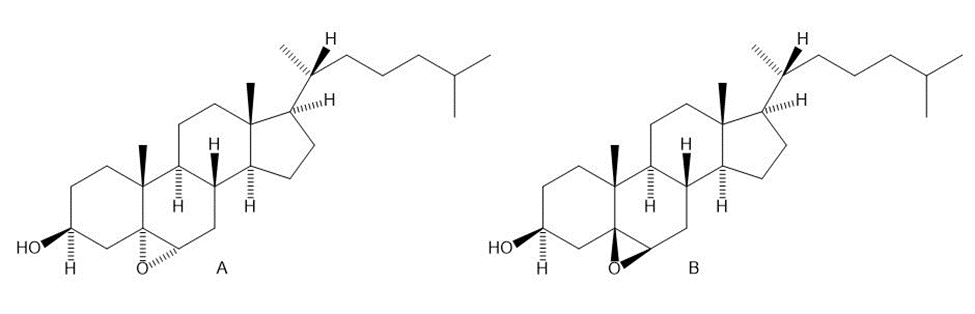
Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the stereochemistry drawn. Why isn’t the isomeric epoxide B formed to any extent?
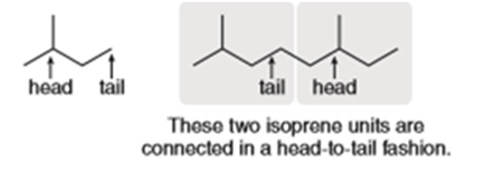
Question: An isoprene unit can be thought of as having a head and a tail. The “head” of the isoprene unit is located at the end of the chain nearest the branch point, and the “tail” is located at the end of the carbon chain farthest from the branch point. Most isoprene units are connected together in a “head-to-tail” fashion, as illustrated. For both lycopene (Problem 31.26), and squalene (Figure 31.9), decide which isoprene units are connected in a head-to-tail fashion and which are not.
Draw the enantiomer and any two diastereomers of cholesterol. Does the OH group of cholesterol occupy an axial or equatorial position?
What do you think about this solution?
We value your feedback to improve our textbook solutions.