Chapter 3: Q.59. (page 91)
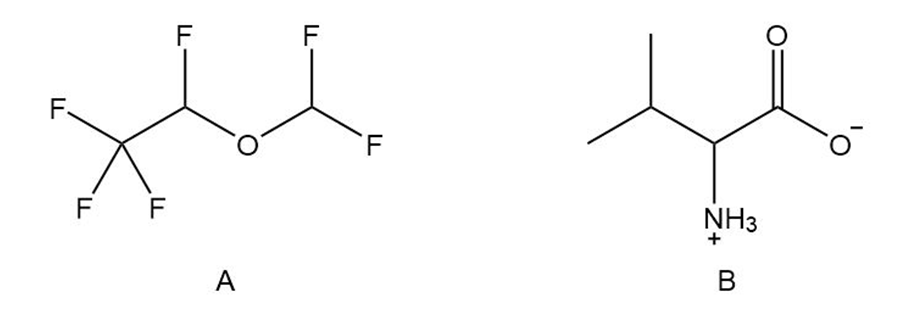
Question: Which compound is more likely to be a general anesthetic? Explain your choice.
Short Answer
Answer
Compound B.
Learning Materials
Features
Discover
Chapter 3: Q.59. (page 91)
Question: Which compound is more likely to be a general anesthetic? Explain your choice.

Answer
Compound B.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:(a)Draw four compounds with molecular formula , each containing at least one different functional group. (b) Predict which compound has the highest boiling point, and explain your reasoning.
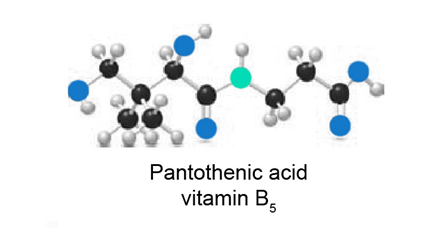
Question:(a) Identify the functional groups in the ball-and-stick model of pantothetic acid, vitamin .
(b) At which sites can pantothenic acid hydrogen bond to water?
(c) Predict the water solubility of pantothenic acid.
Question: Many drugs are sold as their hydrochloride salts , formed by reaction of an amine with HCl.
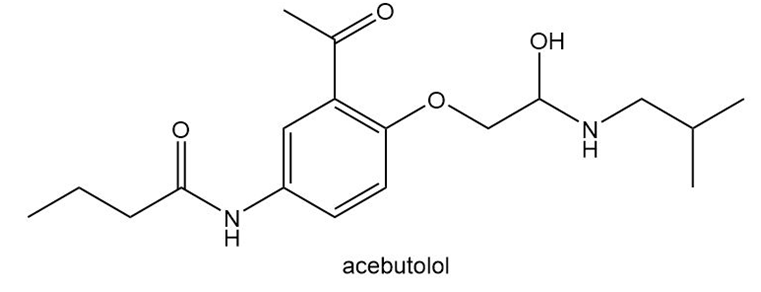
a. Draw the product (a hydrochloride salt) formed by reaction of acebutolol with HCl. Acebutolol is a blocker used to treat high blood pressure. b. Discuss the solubility of acebutolol and its hydrochloride salt in water. c. Offer a reason as to why the drug is marketed as a hydrochloride salt rather than a neutral amine.
Question: By using only electron density arguments, determine whether the following reactions will occur.
a.
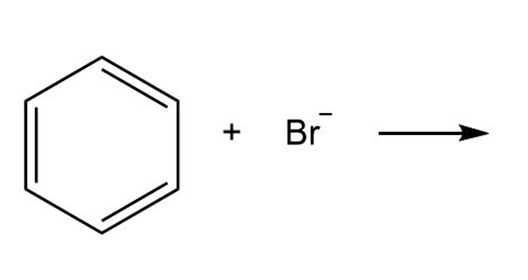
b.
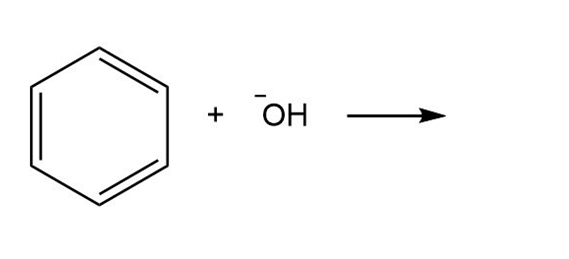
c.
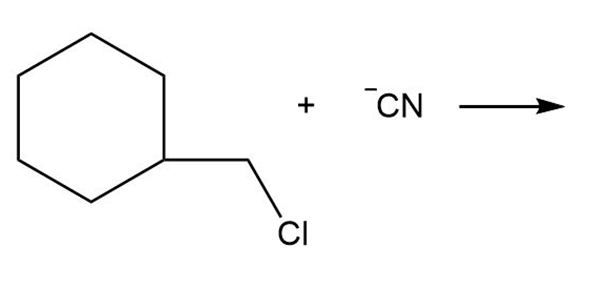
d.
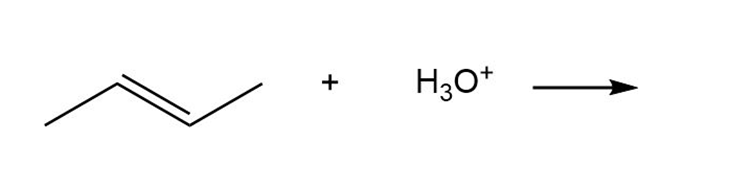
Question: Quinapril (trade name Accupril) is a drug used to treat hypertension and congestive heart failure.
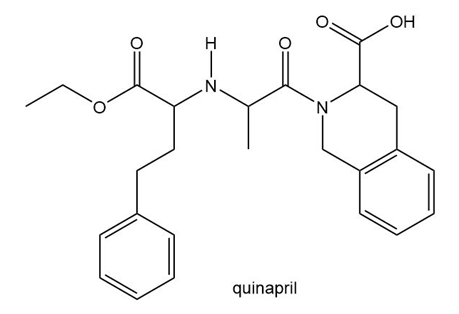
a. Identify the functional groups in quinapril.
b. Classify any alcohol, amide, or amine as , or .
c. At which sites can quinapril hydrogen bond to water?
d. At which sites can quinapril hydrogen bond to acetone ?
e. Label the most acidic hydrogen atom.
f. Which site is most basic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.