Chapter 3: Q.57. (page 91)
Question: By using only electron density arguments, determine whether the following reactions will occur.
a.
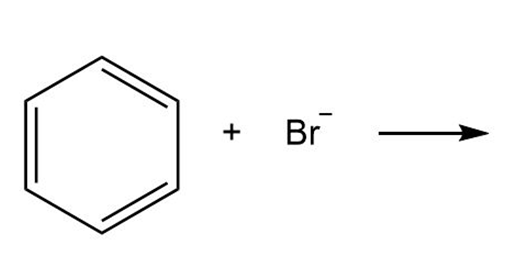
b.
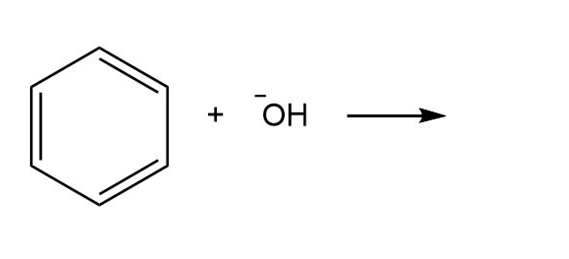
c.
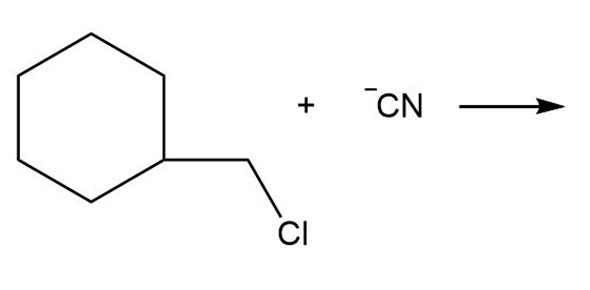
d.
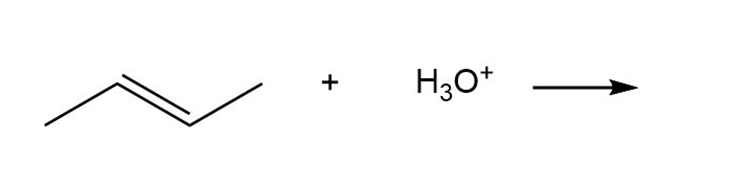
Short Answer
Answer
- No
- No
- Yes
- Yes
Learning Materials
Features
Discover
Chapter 3: Q.57. (page 91)
Question: By using only electron density arguments, determine whether the following reactions will occur.
a.

b.

c.

d.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What types of intermolecular forces are exhibited by each compound?
a.
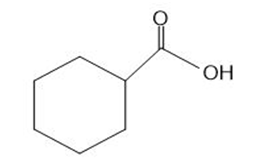
b.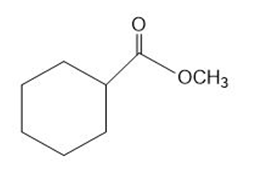
c.
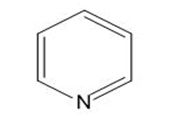
d.
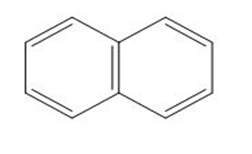
Question: Which of the following molecules can hydrogen bond to another molecule of itself? Which can hydrogen bond with water?
a.
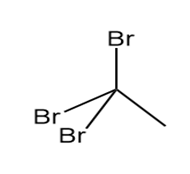
b.
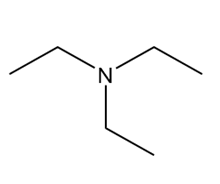
c.
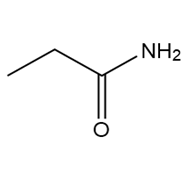
d.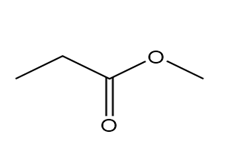
Question: Label the electrophilic and nucleophilic sites in each molecule.
a.
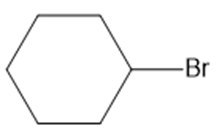
b.
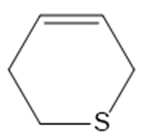
c.
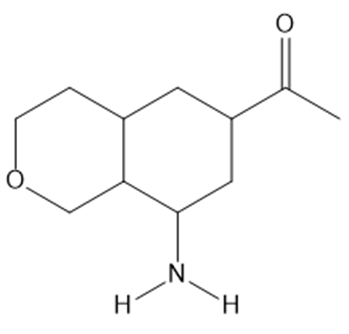
Question: What type of intermolecular forces are present in each compound?
a.
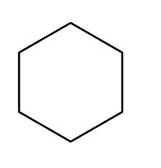
b.
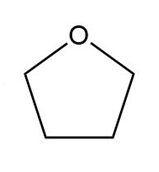
c.
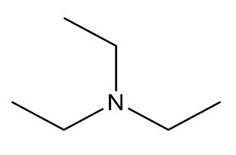
d.
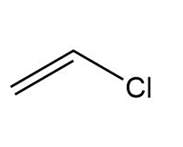
e.
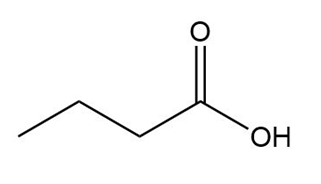
f.
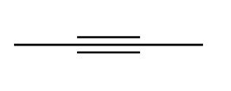
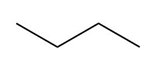
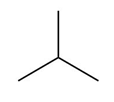
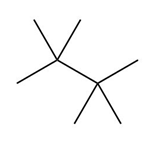
Question:a. Classify the carbon atoms in each compound as ,,, or . b. Classify the hydrogen atoms in each compound as ,,.
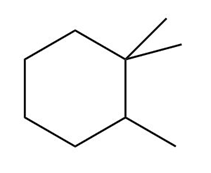
What do you think about this solution?
We value your feedback to improve our textbook solutions.