Chapter 3: Q.46. (page 91)
Question: Rank the following compounds in order of increasing water solubility.
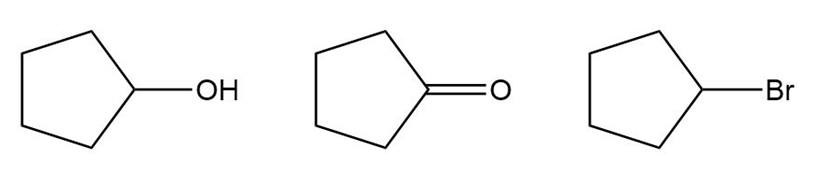
Short Answer
Answer
The compounds can be ranked in the order of increasing water solubility as follows:
Learning Materials
Features
Discover
Chapter 3: Q.46. (page 91)
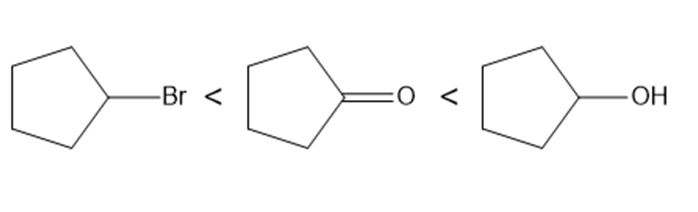
Question: Rank the following compounds in order of increasing water solubility.

Answer
The compounds can be ranked in the order of increasing water solubility as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Label the electrophilic and nucleophilic sites in each molecule.
a.
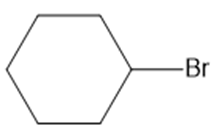
b.
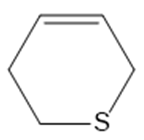
c.
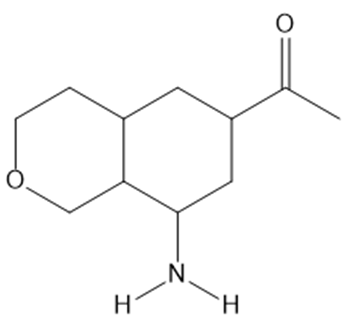
Question: Predict the water solubility of each of the following organic molecules.
a.
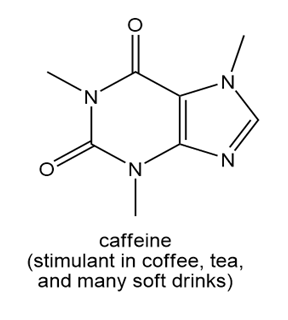
b.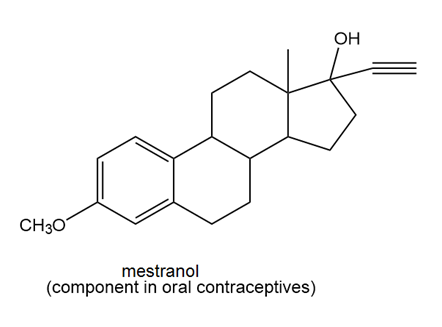
c.
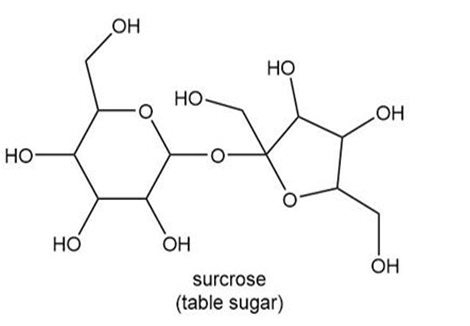
d.
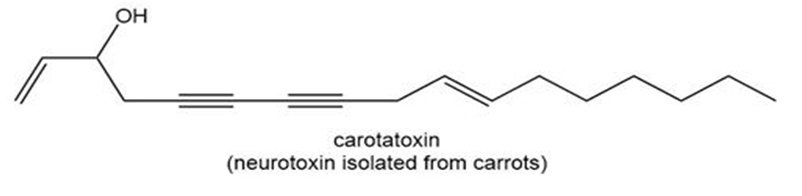
Question: Although diethyl ether and tetrahydrofuran are both four-carbon ethers, one compound is much more water soluble than the other. Predict which compound has higher water solubility and offer an explanation.
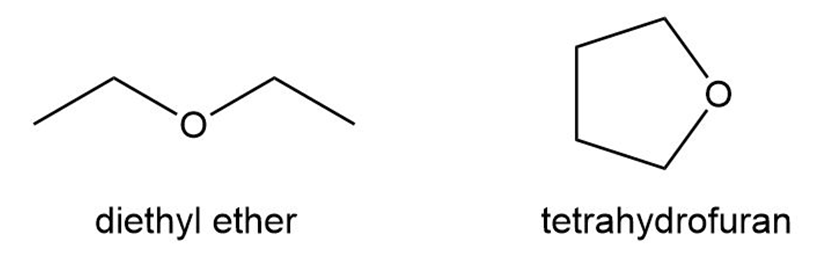
Question: (a)Identify the functional groups in salinosporamide A, an anticancer agent isolated from marine sediment.(b)Classify each alcohol, alkyl halide, amide, and amine as ,, or .
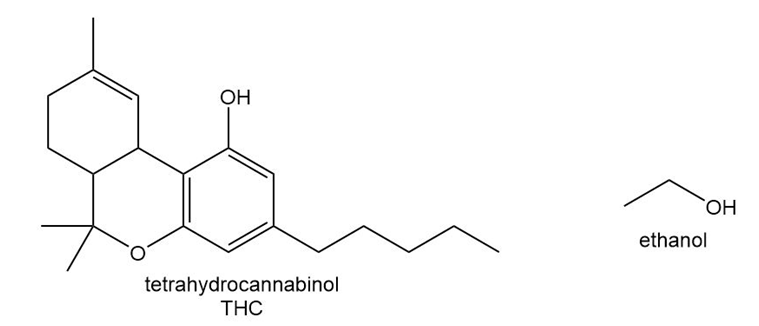
Question: THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body.
What do you think about this solution?
We value your feedback to improve our textbook solutions.