Chapter 3: Q.44. (page 91)
Question: Rank the following compounds in order of increasing melting point.
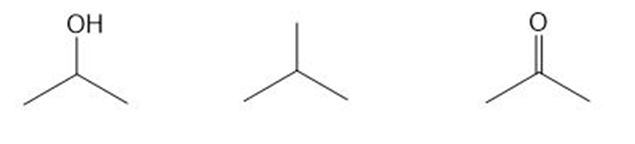
Short Answer
Answer
The compounds can be arranged in the order of increasing melting point as follows:
Learning Materials
Features
Discover
Chapter 3: Q.44. (page 91)
Question: Rank the following compounds in order of increasing melting point.

Answer
The compounds can be arranged in the order of increasing melting point as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Now that you have learned about solubility, explain why aspirin (Section 2.7) crosses a cell membrane as a neutral carboxylic acid rather than an ionic conjugate base.
Question: Explain why diethyl ether and butan-1-ol have similar solubility properties in water, but butan-1-ol has a much higher boiling point.
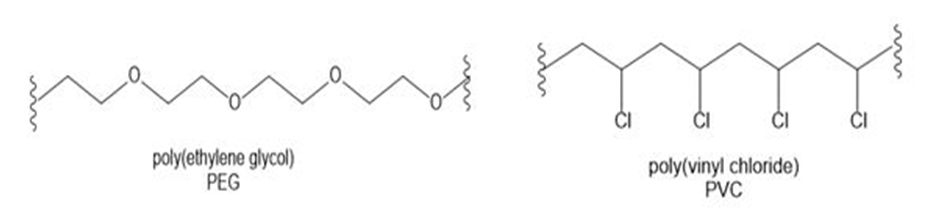
Question: Poly(ethylene glycol) (PEG) and poly(vinyl chloride) (PVC) are examples of polymers, large organic molecules composed of repeating smaller units covalently bonded together. Polymers have very different properties depending (in part) on their functional groups. Discuss the water solubility of each polymer and suggest why PEG is used in shampoos, whereas PVC is used to make garden hoses and pipes. Synthetic polymers are discussed in detail in Chapters 15 and 30.
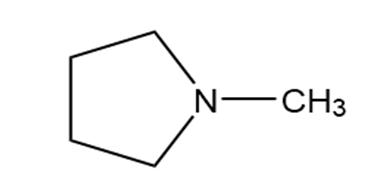
Question: Identify each functional group located in the following rings. Which structure represents a lactone-a cyclic ester- and which represents a lactam-a cyclic amide?
a.
b.
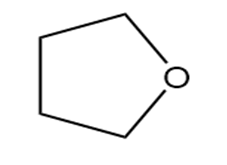
c.
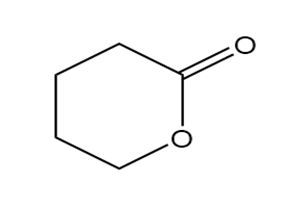
d.
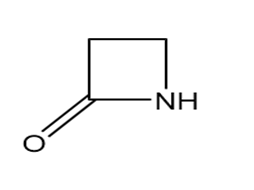
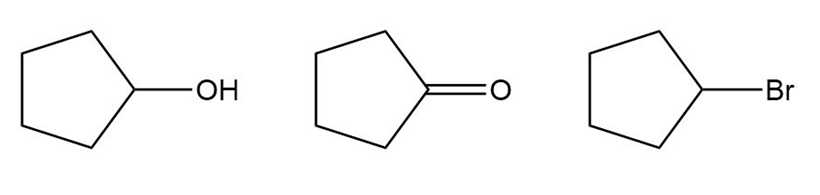
Question: Rank the following compounds in order of increasing water solubility.
What do you think about this solution?
We value your feedback to improve our textbook solutions.