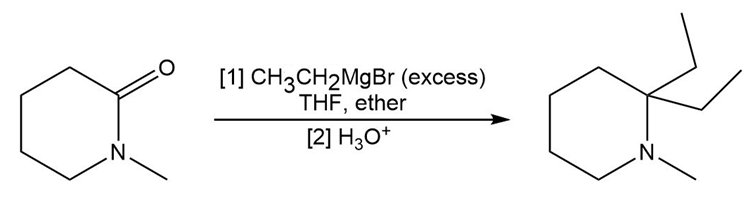
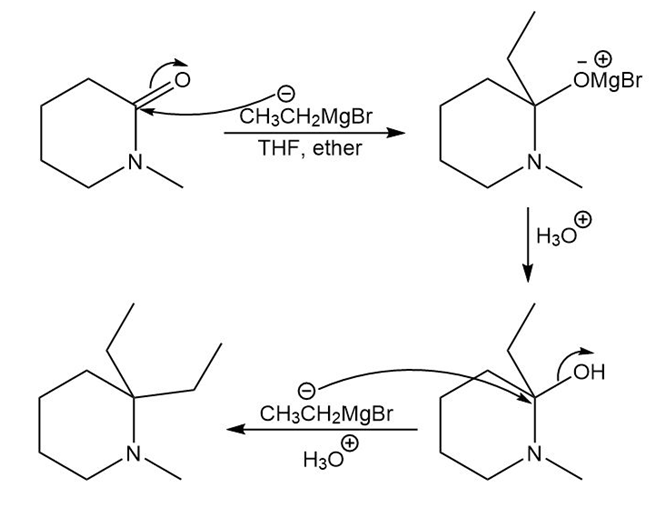
Chapter 20: Q81P (page 764)
Draw a stepwise mechanism for the following reaction of a Grignard reagent with a cyclic amide.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q81P (page 764)
Draw a stepwise mechanism for the following reaction of a Grignard reagent with a cyclic amide.


All the tools & learning materials you need for study success - in one app.
Get started for free
Synthesize each compound from cyclohexanol, ethanol, and any other needed reagents.
a.
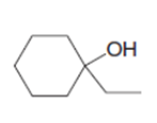
b.
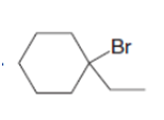
c.

d.
e.
Treatment of isobutene with forms a carbanion that reacts with to form H after water is added to the reaction mixture. H has a molecular ion in its mass spectrum at , and shows fragments at 71 and 68. H exhibits absorptions in its IR spectrum at 3600–3200 and 1651 , and has the NMR spectrum given below. What is the structure of H?
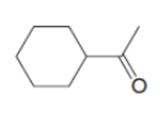
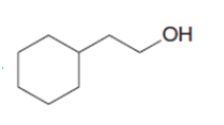
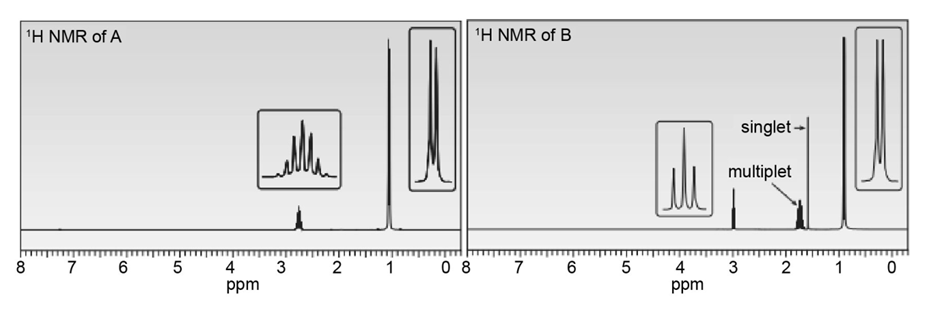
An unknown compound A (molecular formula ) was treated with to form compound B (molecular formula ). Compound A has a strong absorption in its IR spectrum at 1716 . Compound B has a strong absorption in its IR spectrum at 3600–3200 . The NMR spectra of A and B are given. What are the structures of A and B?
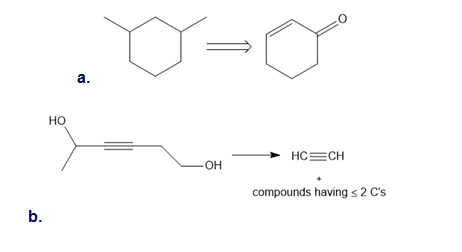
Synthesize each compound from the given starting material. You may use any other required inorganic reagents.
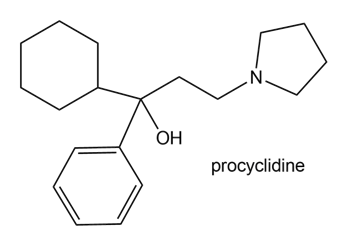
Procyclidine is a drug that has been used to treat the uncontrolled body movements associated with Parkinson’s disease. Draw three different methods to prepare procyclidine using a Grignard reagent
What do you think about this solution?
We value your feedback to improve our textbook solutions.