Chapter 20: Q35 (page 805)
Synthesize each compound from cyclohexanol, ethanol, and any other needed reagents.
a.
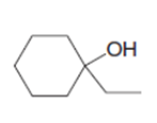
b.
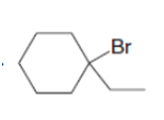
c.
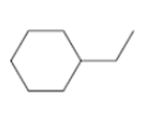
d.
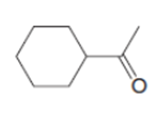
e.
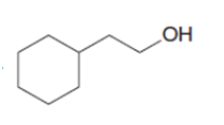
Short Answer
The given products can be synthesized through the following method:
a.
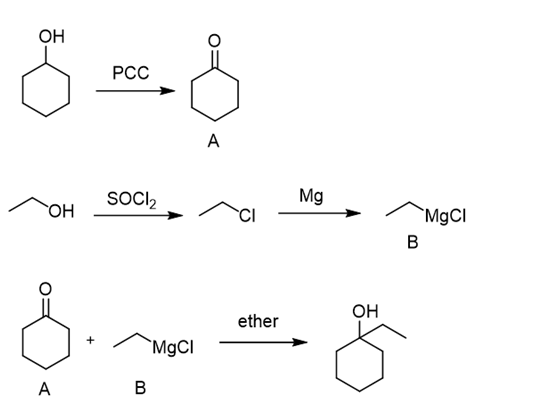
b.
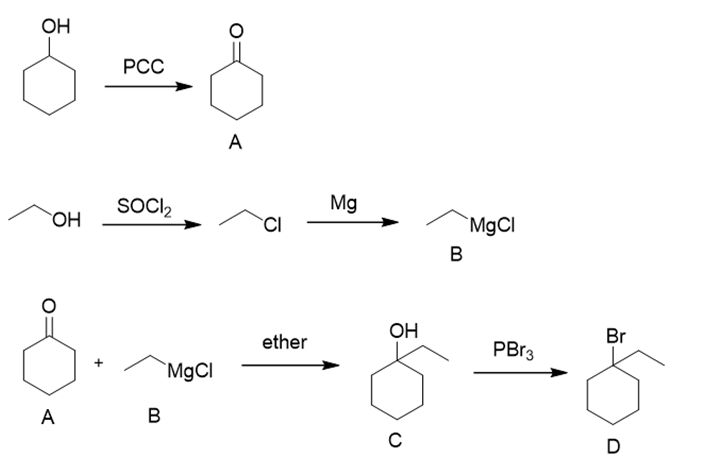
c.
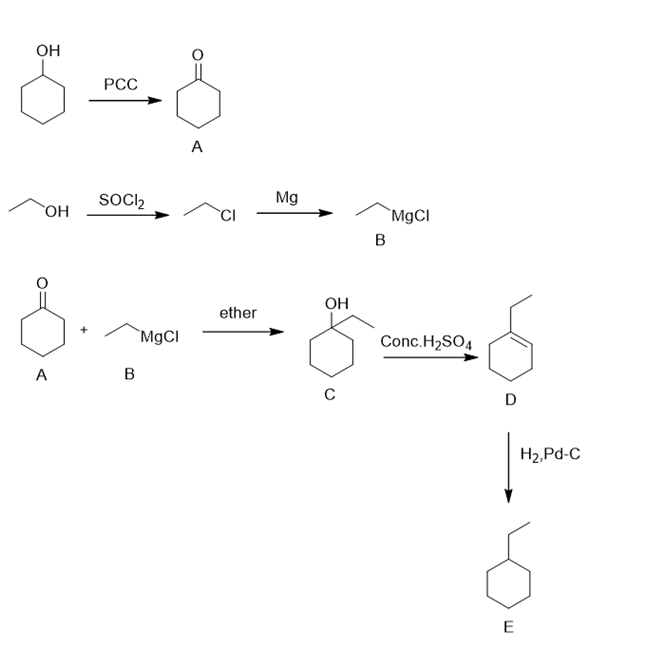
d.
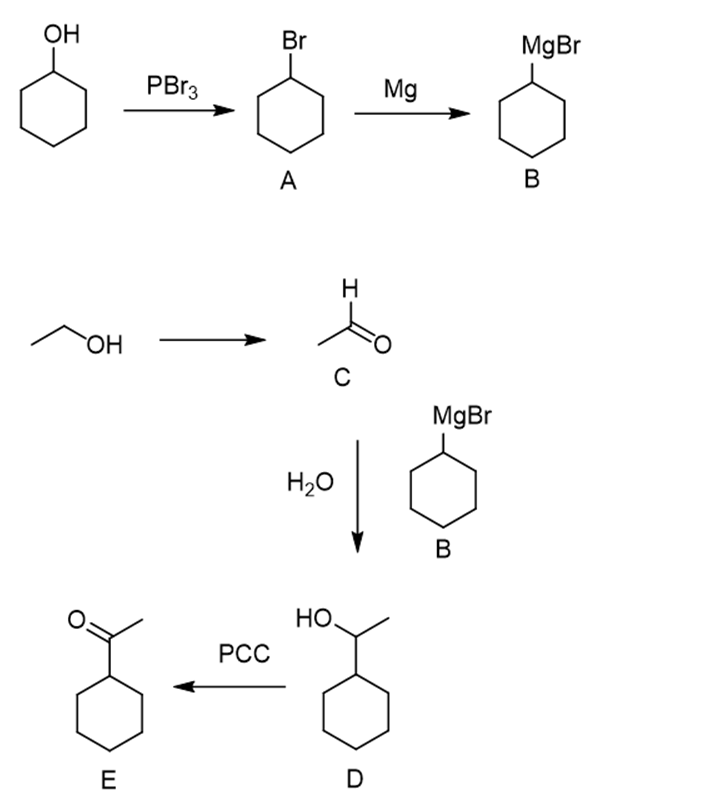
e.
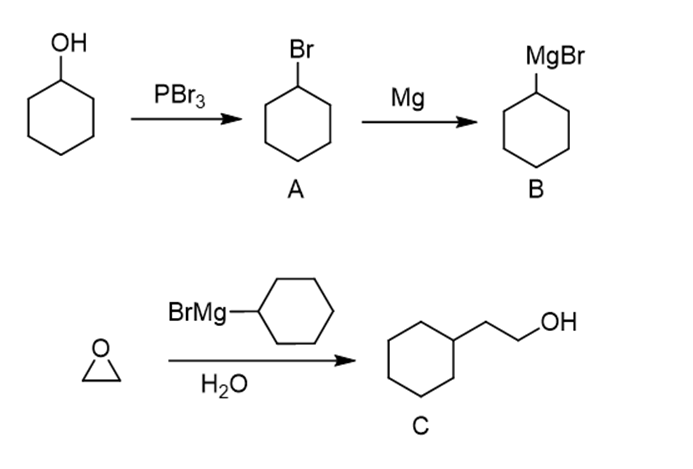
Learning Materials
Features
Discover
Chapter 20: Q35 (page 805)
Synthesize each compound from cyclohexanol, ethanol, and any other needed reagents.
a.

b.

c.

d.

e.

The given products can be synthesized through the following method:
a.

b.

c.

d.

e.

All the tools & learning materials you need for study success - in one app.
Get started for free
Synthesize each compound from cyclohexanol using any other organic or inorganic compounds.
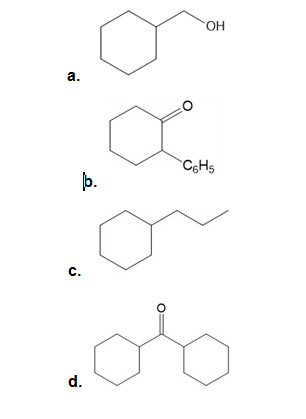
Treatment of isobutene with forms a carbanion that reacts with to form H after water is added to the reaction mixture. H has a molecular ion in its mass spectrum at , and shows fragments at 71 and 68. H exhibits absorptions in its IR spectrum at 3600–3200 and 1651 , and has the NMR spectrum given below. What is the structure of H?
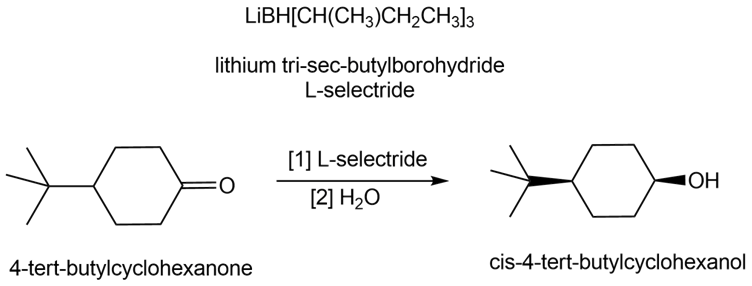
Lithium tri-sec-butylborohydride, also known as L-selectride, is a metal hydride reagent that contains three sec-butyl groups bonded to boron. When this reagent is used to reduce cyclic ketones, one stereoisomer often predominates as product. Explain why the reduction of 4-tert-butylcyclohexanone with L-selectride forms the cis alcohol as the major product.
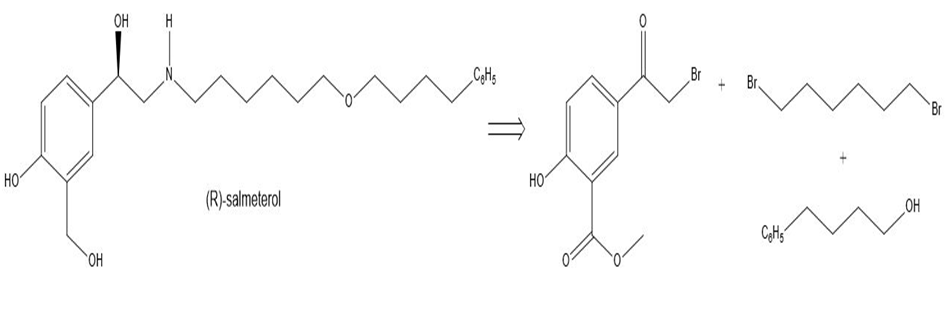
Design a synthesis of (R)-salmeterol (Figure 20.3) from the following starting materials.
stereochemistry of the products of reduction depends on the reagent used,as you learned in Sections 20.5 and 20.6. With this in mind, how would you convert3,3-dimethylbutan-2-one to: (a) racemic 3,3-dimethylbutan-2-ol ;(b) only (R)-3,3-dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.