Chapter 20: Q59 (page 812)
What epoxide and organometallic reagents are needed to synthesize each alcohol?
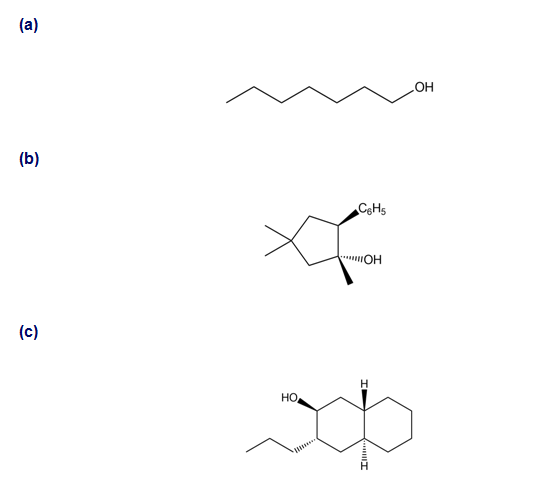
Short Answer
a.

b.
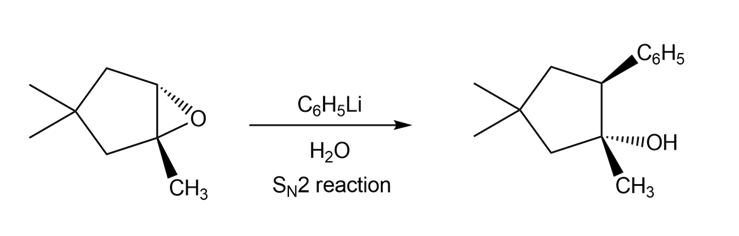
c.
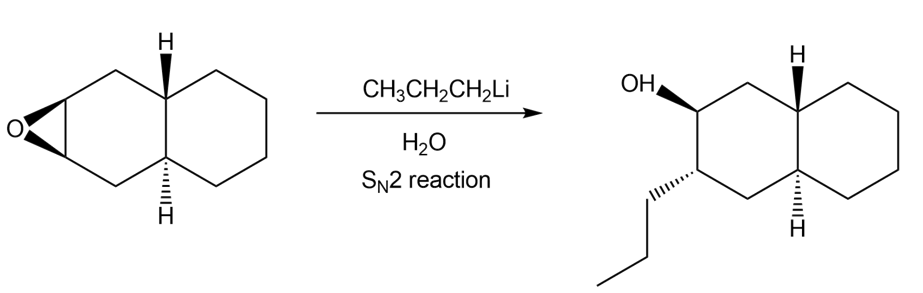
Learning Materials
Features
Discover
Chapter 20: Q59 (page 812)
What epoxide and organometallic reagents are needed to synthesize each alcohol?

a.

b.

c.
All the tools & learning materials you need for study success - in one app.
Get started for free
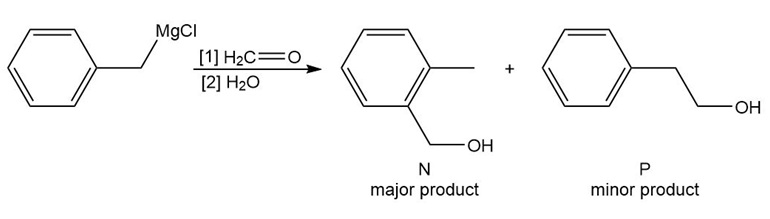
Reaction of benzyl magnesium chloride with formaldehyde yields alcohols N and P after protonation. Draw a stepwise mechanism that shows how both products are formed.
Propose two different methods to synthesize oct-1-en-3-ol using a Grignard reagent and a carbonyl compound. Oct-1-en-3-ol is commonly called matsutake alcohol because it was first isolated from the Japanese matsutake mushroom.
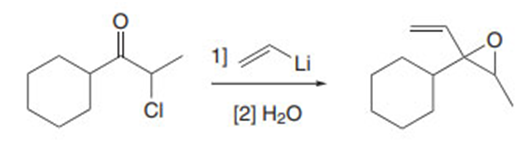
Draw a stepwise mechanism for the following reaction.
Treatment of compound C (molecular formula ) with , followed by H2O, affords compound D (molecular formula ). Compound D has a strong peak in its IR spectrum at 3600–3200 . The NMR spectral data of C and D are given. What are the structures of C and D?Compound C signals at 1.3 (singlet, 6 H) and 2.4 (singlet, 2 H) ppm Compound D signals at 1.2 (singlet, 6 H), 1.6 (singlet, 1 H), 2.7 (singlet, 2 H), and 7.2 (multiplet, 5 H) ppm
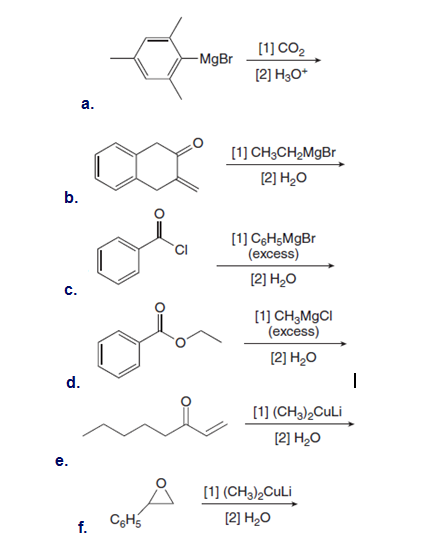
Draw the products of the following reactions with organometallic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.