Chapter 20: Q79 (page 816)
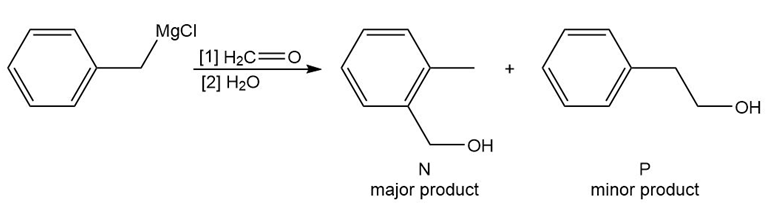
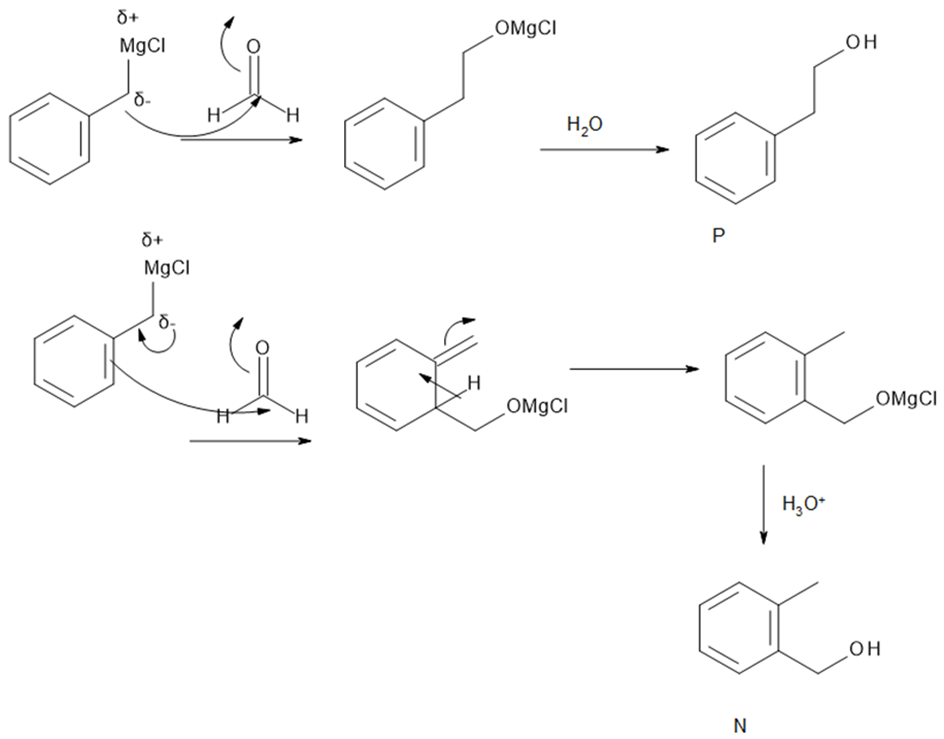
Reaction of benzyl magnesium chloride with formaldehyde yields alcohols N and P after protonation. Draw a stepwise mechanism that shows how both products are formed.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q79 (page 816)
Reaction of benzyl magnesium chloride with formaldehyde yields alcohols N and P after protonation. Draw a stepwise mechanism that shows how both products are formed.


All the tools & learning materials you need for study success - in one app.
Get started for free
Design a synthesis of each compound from alcohols having four carbons or fewer as the only organic starting materials. You may use any other inorganic reagents you choose.
Treatment of compound E (molecular formula ) with excess yieldscompound F (molecular formula ) after protonation with . E shows a strongabsorption in its IR spectrum at 1743. F shows a strong IR absorption at 3600–3200.The NMR spectral data of E and F are given.
What are the structures of E and F?
Compound E signals at 1.2 (triplet, 3 H), 2.0 (singlet, 3 H), and 4.1 (quartet, 2 H) ppm
Compound F signals at 0.9 (triplet, 6 H), 1.1 (singlet, 3 H), 1.5 (quartet, 4 H), and 1.55(singlet, 1 H) ppm
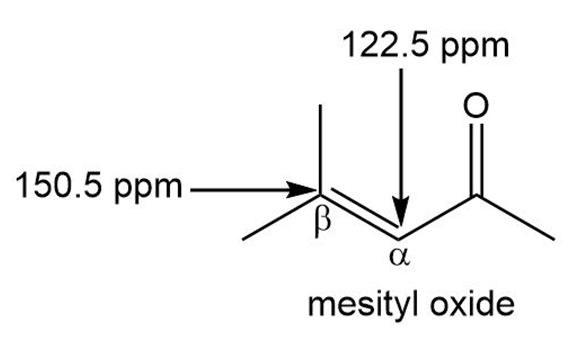
Explain why the carbon of an ,-unsaturated carbonyl compound absorbs farther downfield in the NMR spectrum than the a carbon, even though the carbon is closer to the electron-withdrawing carbonyl group. For example, the carbon of mesityl oxide absorbs at 150.5 ppm, while the carbon absorbs at 122.5 ppm.
stereochemistry of the products of reduction depends on the reagent used,as you learned in Sections 20.5 and 20.6. With this in mind, how would you convert3,3-dimethylbutan-2-one to: (a) racemic 3,3-dimethylbutan-2-ol ;(b) only (R)-3,3-dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
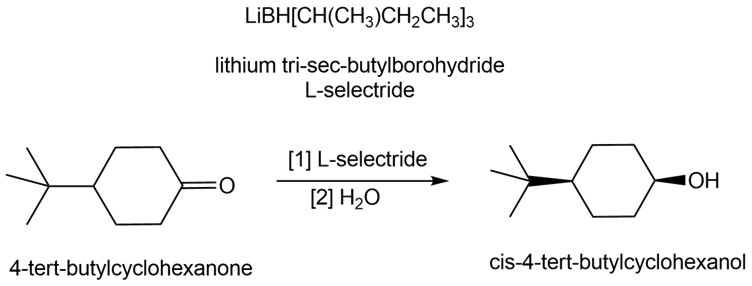
Lithium tri-sec-butylborohydride, also known as L-selectride, is a metal hydride reagent that contains three sec-butyl groups bonded to boron. When this reagent is used to reduce cyclic ketones, one stereoisomer often predominates as product. Explain why the reduction of 4-tert-butylcyclohexanone with L-selectride forms the cis alcohol as the major product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.