Chapter 20: Q39 (page 809)
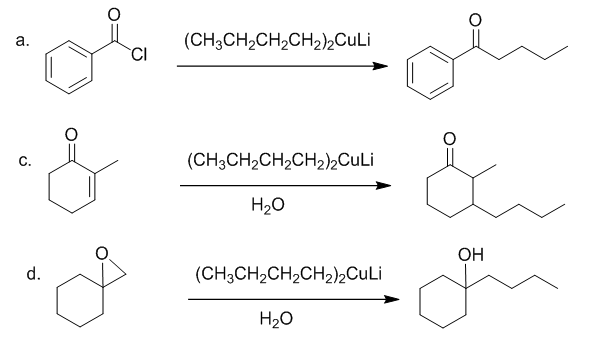
Draw the product formed when is treated with each compound. In some cases, no reaction occurs.
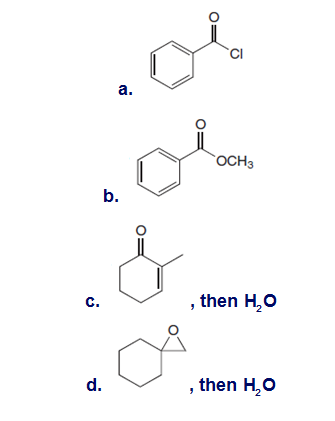
Short Answer
The products formed when is treated with each compound are shown below:
Learning Materials
Features
Discover
Chapter 20: Q39 (page 809)
Draw the product formed when is treated with each compound. In some cases, no reaction occurs.

The products formed when is treated with each compound are shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
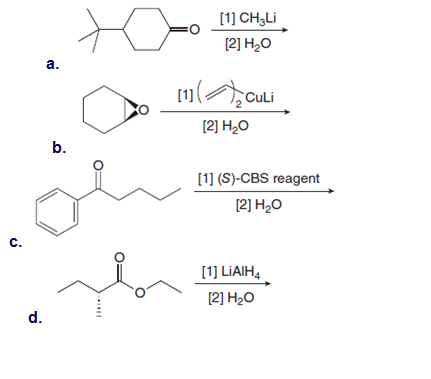
Draw all stereoisomers formed in each reaction.
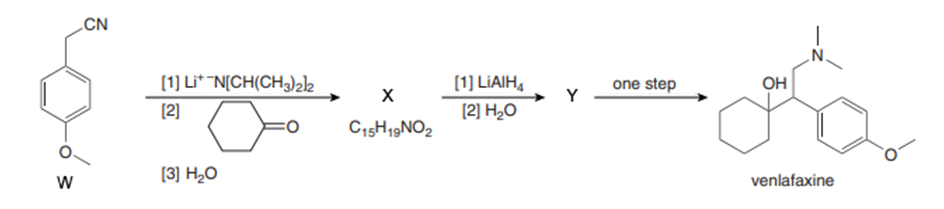
Identify X and Y, two of the intermediates in a synthesis of the antidepressant venlafaxine (trade name Effexor), in the following reaction scheme. Write a mechanism for the formation of X from W.
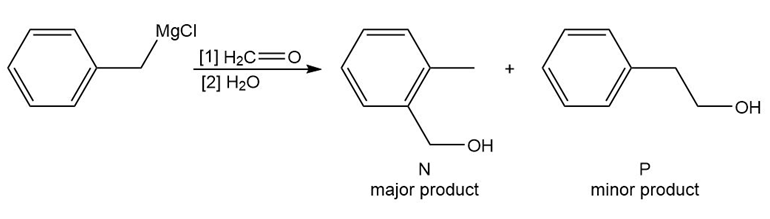
Reaction of benzyl magnesium chloride with formaldehyde yields alcohols N and P after protonation. Draw a stepwise mechanism that shows how both products are formed.
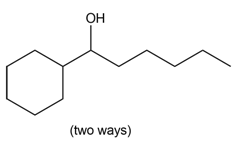
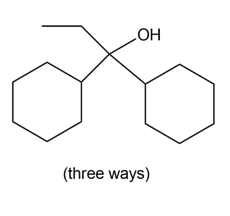
What organolithium reagent and carbonyl compound can be used to prepare each of the following compounds? You may use aldehydes, ketones, or esters as carbonyl starting materials.
a
b.
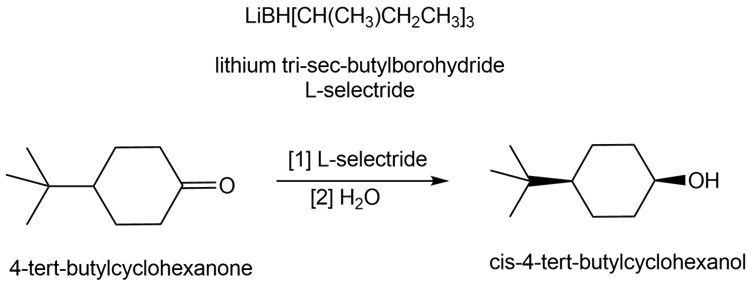
Lithium tri-sec-butylborohydride, also known as L-selectride, is a metal hydride reagent that contains three sec-butyl groups bonded to boron. When this reagent is used to reduce cyclic ketones, one stereoisomer often predominates as product. Explain why the reduction of 4-tert-butylcyclohexanone with L-selectride forms the cis alcohol as the major product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.