Chapter 16: PROBLEM 16.62 (page 638)
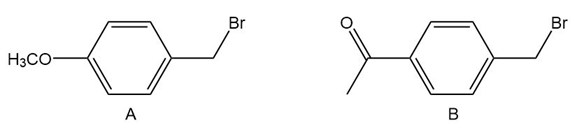
Question: Which benzylic halide reacts faster in an SN1 reaction? Explain
Short Answer
Answer
Compound A
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.62 (page 638)
Question: Which benzylic halide reacts faster in an SN1 reaction? Explain

Answer
Compound A
All the tools & learning materials you need for study success - in one app.
Get started for free
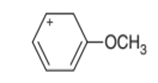
Question. Draw all possible resonance structures for the following cation and indicate which structure makes the largest contribution to the resonance hybrid.
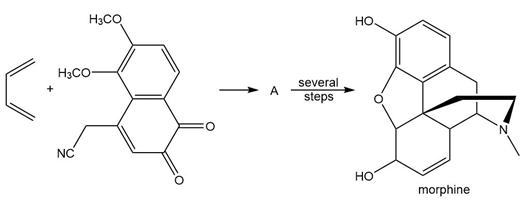
Question: Draw the product (A) of the following Diels-Alder reaction. A was a key intermediate in the synthesis of the addicting pain reliever morphine, the chapter-opening molecule.
Question: Draw the products formed when each diene is treated with one equivalent of HCl.
a.
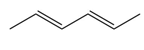
b.
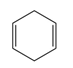
c.
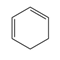
d.
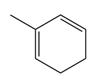
Question: Neuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent.
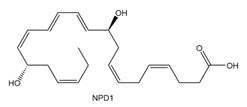
a. Label each double bond as conjugated or isolated.
b. Label each double bond as E or Z.
c. For each conjugated system, label the given conformation as s-cis or s-trans.
Question: Addition of HCl to alkene X forms two alkyl halides Y and Z.
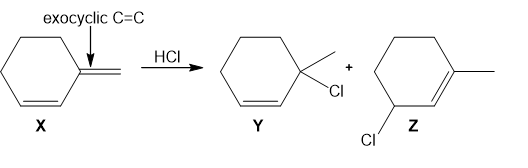
a. Label Y and Z as a 1,2-addition product or a 1,4-addition product.
b. Label Y and Z as the kinetic or thermodynamic product and explain why.
c. Explain why addition of HCl occurs at the indicated C=C(called an exocyclic double bond), rather than the other C=C (called an endocyclic double bond).
What do you think about this solution?
We value your feedback to improve our textbook solutions.