Chapter 16: PROBLEM 16.52 (page 637)
Question: Compounds containing triple bonds are also Diels–Alder dienophiles. With this in mind, draw the products of each reaction.
a.
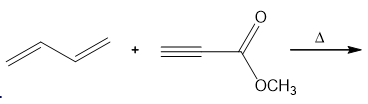
b.
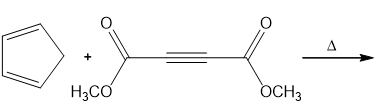
Short Answer
a.
 Product of a.
Product of a.
b.
 Product of b.
Product of b.
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.52 (page 637)
Question: Compounds containing triple bonds are also Diels–Alder dienophiles. With this in mind, draw the products of each reaction.
a.
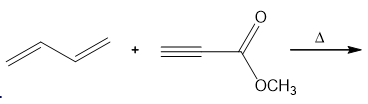
b.

a.
 Product of a.
Product of a.
b.
 Product of b.
Product of b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Why is the bond dissociation energy for the C-C bond in ethane much higher than the bond dissociation energy for the labeled C-C in but-1-ene?
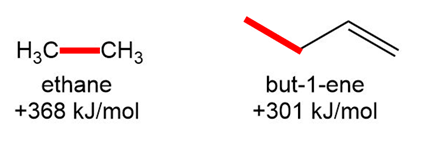
Question: Use resonance theory and the Hammond postulate to explain why 3-chloroprop-1-ene is more reactive than 1-chloropropane in SN1 reactions.
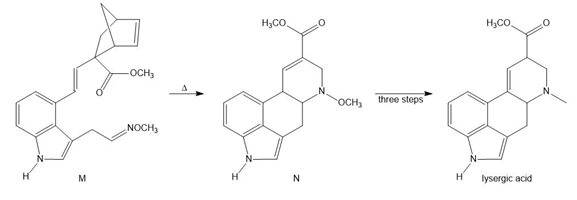
Question: Devise a stepwise mechanism for the conversion of M to N. N has been converted in several steps to lysergic acid, a naturally occurring precursor of the hallucinogen LSD (Figure 18.4).
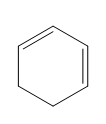
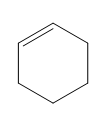
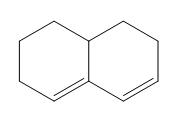
Question: Classify each diene as isolated or conjugated.
a.
b.
c.
d.
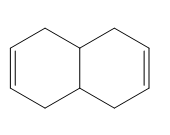
Question: What diene and dienophile are needed to prepare each product?
a.
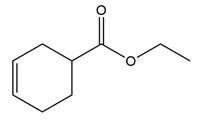
b.
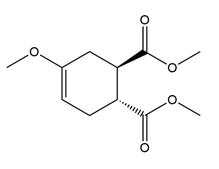
c.
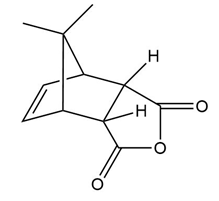
What do you think about this solution?
We value your feedback to improve our textbook solutions.