Chapter 16: PROBLEM 16.27 (page 632)
Question: Which compound in each pair absorbs UV light at a longer wavelength?
a.
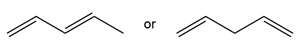
b.
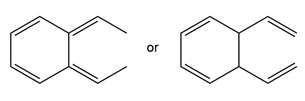
Short Answer
Answer
a.
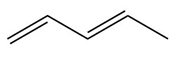
b.
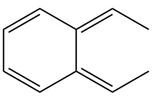
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.27 (page 632)
Question: Which compound in each pair absorbs UV light at a longer wavelength?
a.
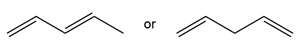
b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following molecules absorbs a longer wavelength of UV light?

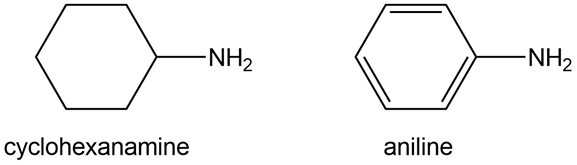
Question: Determine the hybridization around the N atom in each amine and explain why cyclohexanamine is 106 times more basic than aniline
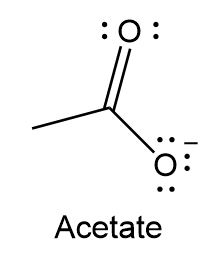
Question: Use resonance theory explains why the labeled C-O bond lengths are equal in the acetate ion.
Question: Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.
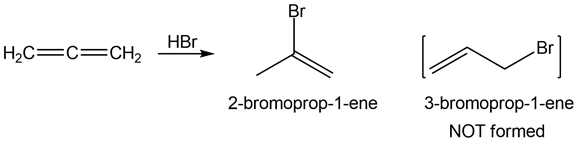
Question: Addition of HBr to alleneforms 2-bromoprop-1-ene rather than 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed from an allylic carbocation. Considering the arrangement of orbitals in the allene reactant, explain this result.
What do you think about this solution?
We value your feedback to improve our textbook solutions.