Chapter 22: Q46. (page 915)

Short Answer
Answer
Learning Materials
Features
Discover
Chapter 22: Q46. (page 915)

Answer


All the tools & learning materials you need for study success - in one app.
Get started for free
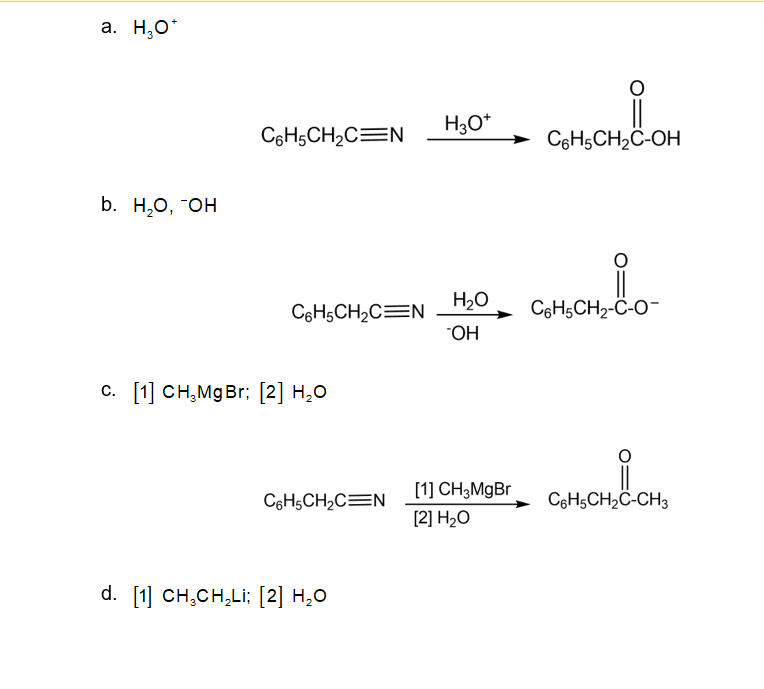
What Grignard reagent and aldehyde (or ketone) are needed to prepare each alcohol? Show all possible routes
(a)
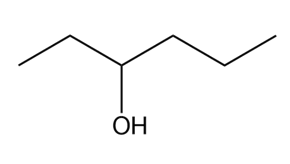
(b)
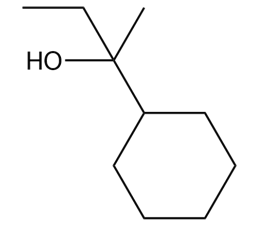
(c)
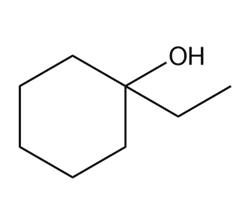
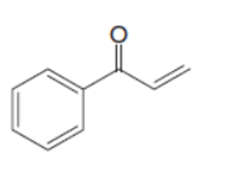
Question: Draw a tautomer of each compound.
a.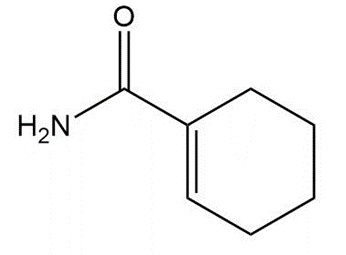
b.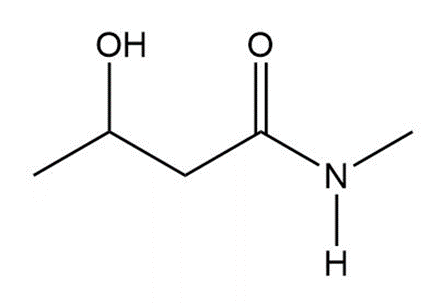
c. 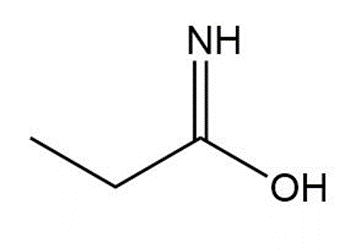
Tertiary alcohols can be formed by the reaction of dimethyl carbonate [(CH3O)2CO ] with excess Grignard reagent. Draw a stepwise mechanism for the following transformation.
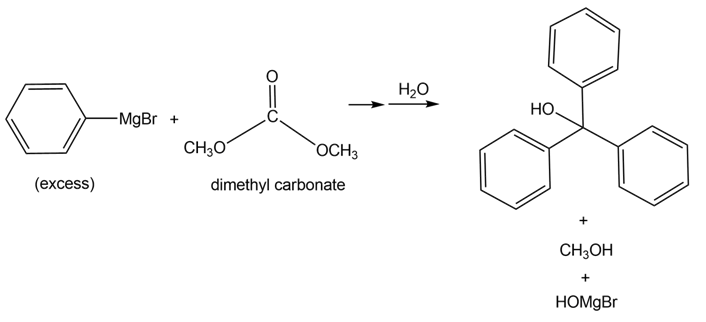
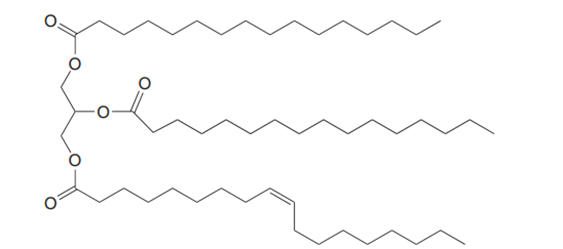
Question: What is the composition of the soap prepared by hydrolysis of the following triacylglycerol?
Draw the product when each compound is treated with either (CH3)2CuLi , followed by H2O , HCCLi , followed by H2O .
a.
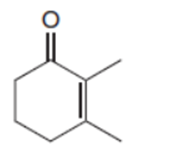
b.
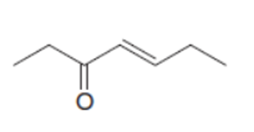
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.