Chapter 19: Q28. (page 757)
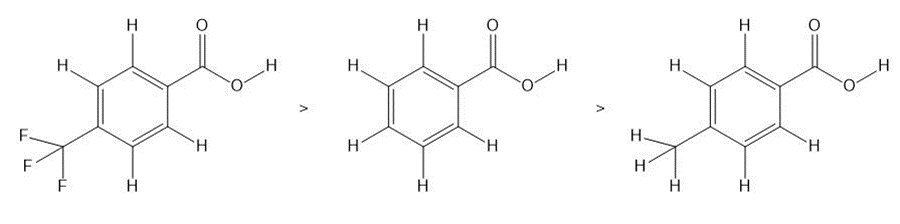
Question: Rank the carboxylic acids in order of increasing acidity.
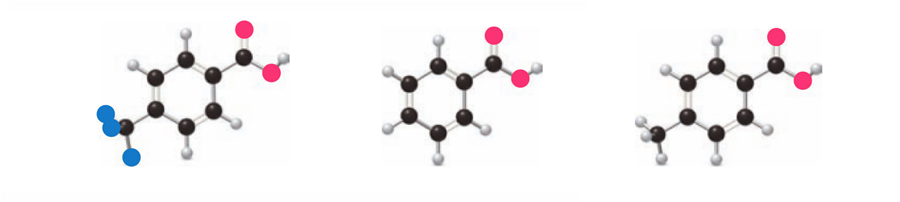
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 19: Q28. (page 757)
Question: Rank the carboxylic acids in order of increasing acidity.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain why amino acids, unlike most other organic compounds, are insoluble in organic solvents like diethyl ether.
Question: Two other commonly used sulfonic acids are methanesulfonic acid and trifluoromethanesulfonic acid . Which has the weaker conjugate base? Which conjugate base is the better leaving group? Which of these acids has the higher ?
Can octane and octan-1-ol be separated using an aqueous extraction procedure? Explain why or why not.
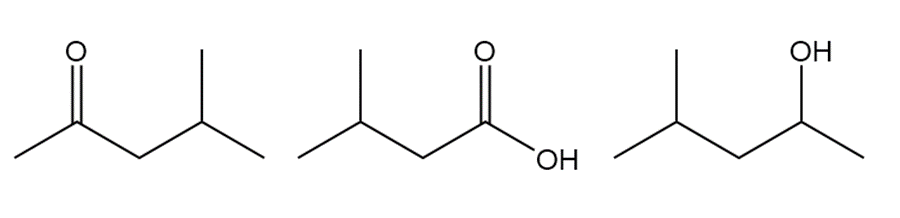
Question: Rank the following compounds in order of increasing boiling point.
Question: Give the IUPAC name for each compound.
a. 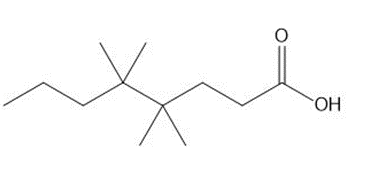
b.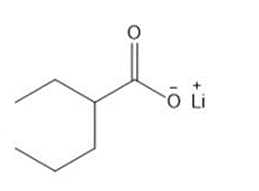
c. 
d.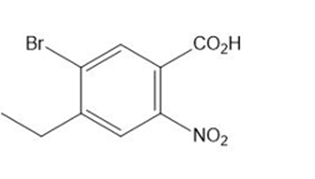
e.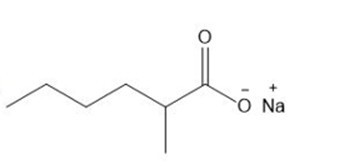
f.
What do you think about this solution?
We value your feedback to improve our textbook solutions.