Chapter 19: Q22. (page 753)
Question: Draw both enantiomers of each amino acid and label them as Ror S:(a) phenylalanine; (b) methionine.
Short Answer
Answer
a.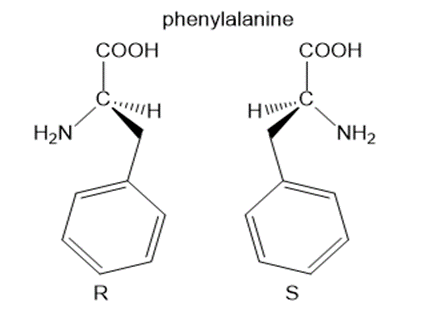
b.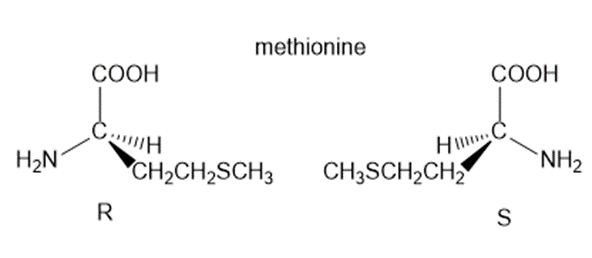
Learning Materials
Features
Discover
Chapter 19: Q22. (page 753)
Question: Draw both enantiomers of each amino acid and label them as Ror S:(a) phenylalanine; (b) methionine.
Answer
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate the isoelectric point for each amino acid.
a. cysteine: (COOH) = 2.05; =10.25
b. methionine: (COOH) = 2.28; =9.21
Question: Depakote, a drug used to treat seizures and bipolar disorder, consists of a mixture of valproic acid and its sodium salt. Give IUPAC names for each of these compounds.
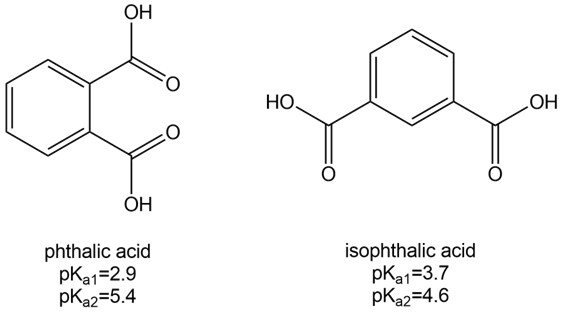
Phthalic acid and isophthalic acid have protons on two carboxy groups that can be removed with base.
(a) Explain why the for loss of the first proton () is lower for phthalic acid than isophthalic acid.
(b) Explain why the for loss of the second proton ( ) is higher for phthalic acid than isophthalic acid.
Draw all resonace strucutres of the conjugate bases formed by removal of the labeled protons in cyclohexane-1,3-dione and acetanilide. For each compound, rank these protons in order of increasing acidity and explain the order you chose.
a.

b.

Question: Two other commonly used sulfonic acids are methanesulfonic acid and trifluoromethanesulfonic acid . Which has the weaker conjugate base? Which conjugate base is the better leaving group? Which of these acids has the higher ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.