Chapter 17: Q.23. (page 641)
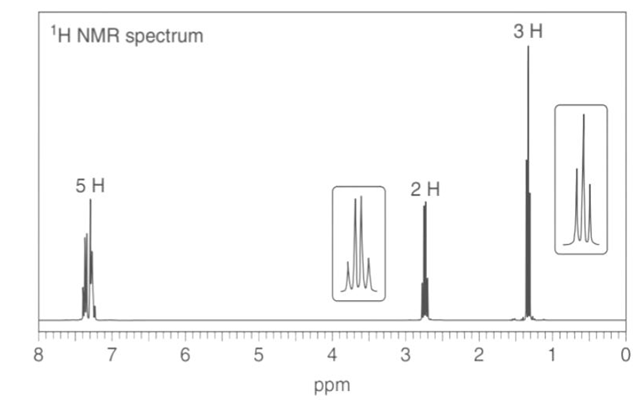
Question: Name each compound and state how many lines are observed in its NMR spectrum.
Short Answer
Answer
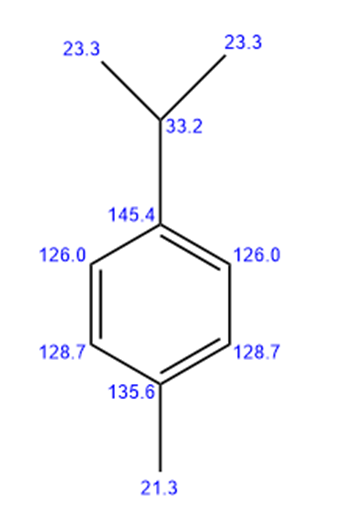
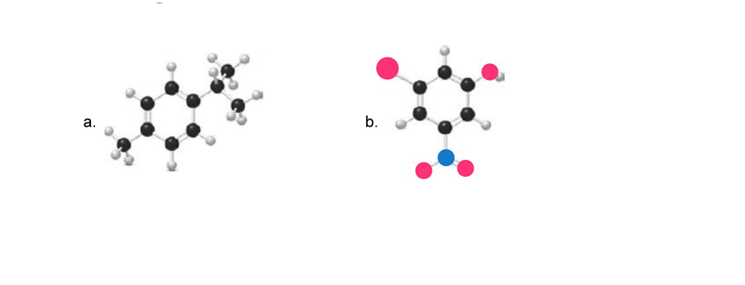
a.
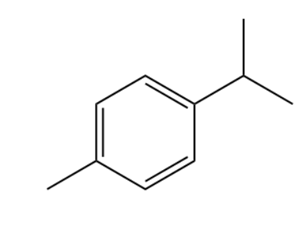
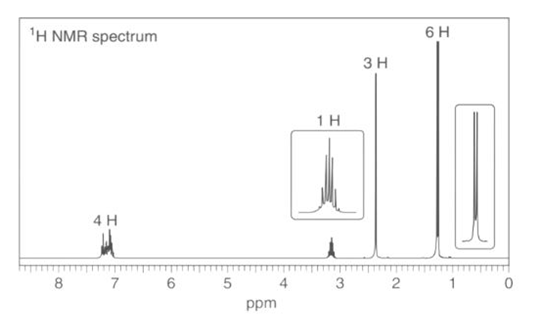
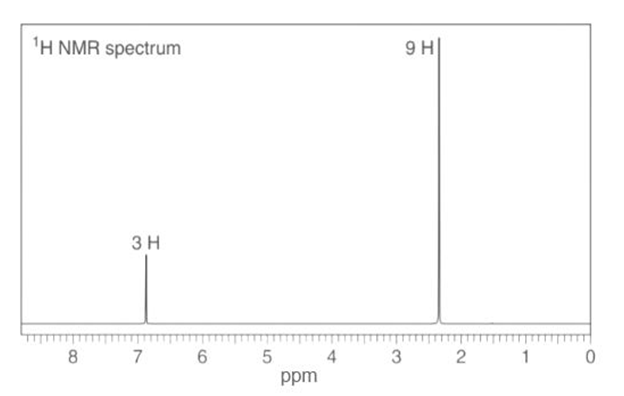
4-methyl isopropyl benzene
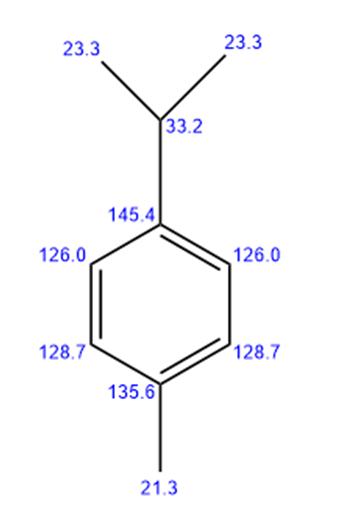
Seven lines for the above structure
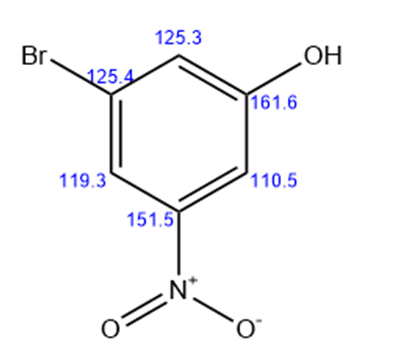
b.
3-nitro-5-bromo phenol
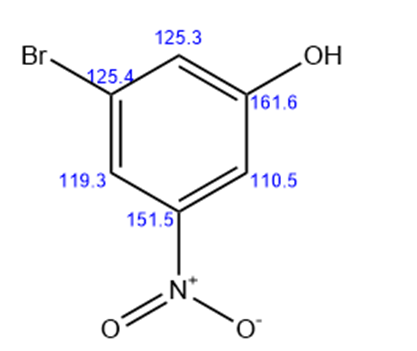
Six lines for the above structure