Chapter 29: Q44P (page 1193)
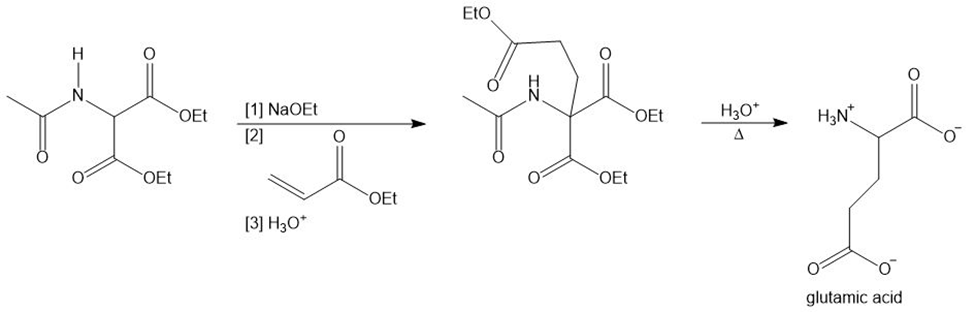
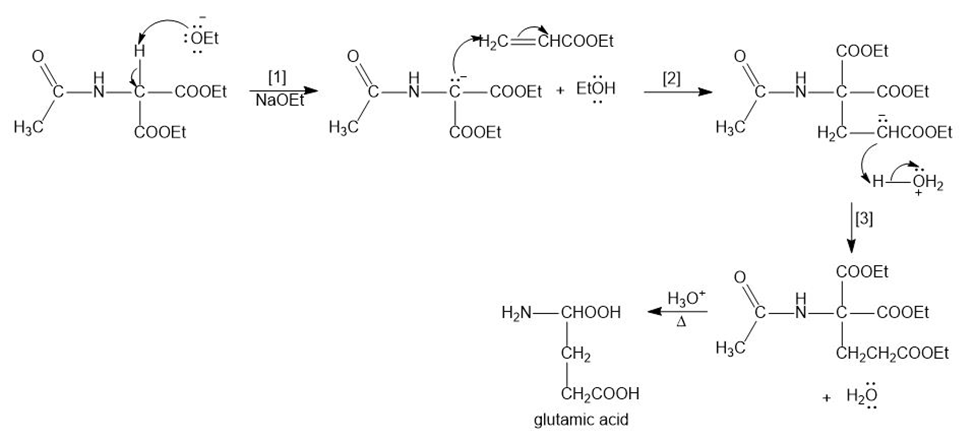
Glutamic acid is synthesized by the following reaction sequence. Draw a stepwise mechanism for Steps [1]–[3].
Short Answer
Learning Materials
Features
Discover
Chapter 29: Q44P (page 1193)
Glutamic acid is synthesized by the following reaction sequence. Draw a stepwise mechanism for Steps [1]–[3].


All the tools & learning materials you need for study success - in one app.
Get started for free
Deduce the sequence of a heptapeptide that contains the amino acids Ala, Arg, Glu, Gly, Leu, Phe, and Ser, from the following experimental data. Edman degradation cleaves Leu from the heptapeptide, and carboxypeptidase forms Glu and a hexapeptide. Treatment of the heptapeptide with chymotrypsin forms a hexapeptide and a single amino acid. Treatment of the heptapeptide with trypsin forms a pentapeptide and a dipeptide. Partial hydrolysis forms Glu, Leu, Phe, and the tripeptidesGly–Ala–Ser and Ala–Ser–Arg
Histidine is classified as a basic amino acid because one of the N atoms in its five-membered ring is readily protonated by acid. Which N atom in histidine is protonated and why?
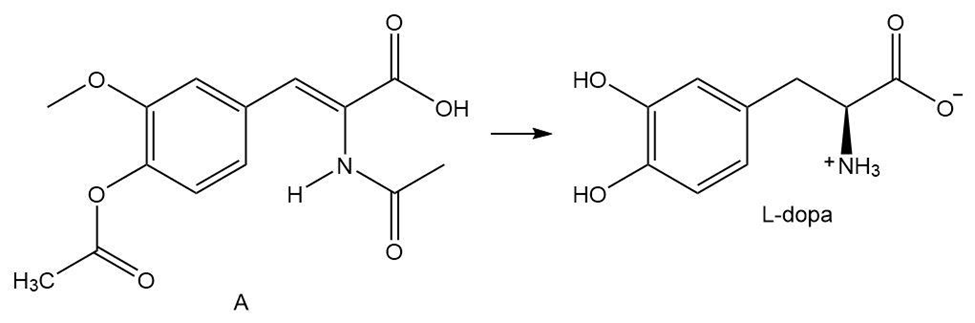
What steps are needed to convert A to L-dopa, an uncommon amino acid that is effective in treating Parkinson’s disease? These steps are the key reactions in the first commercial asymmetric synthesis using a chiral transition metal catalyst. This process was developed at Monsanto in 1974.
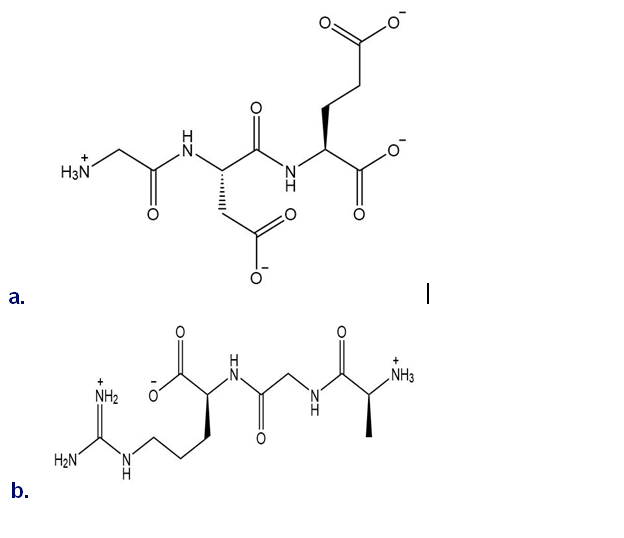
Name each peptide using both the three-letter and one-letter abbreviations of the component amino acids.
What types of stabilizing interactions exist between each of the following pairs of amino acids?
a. Ser and Tyr
b. Val and Leu
c. two Phe residues
What do you think about this solution?
We value your feedback to improve our textbook solutions.