Chapter 29: Q44. (page 1193)
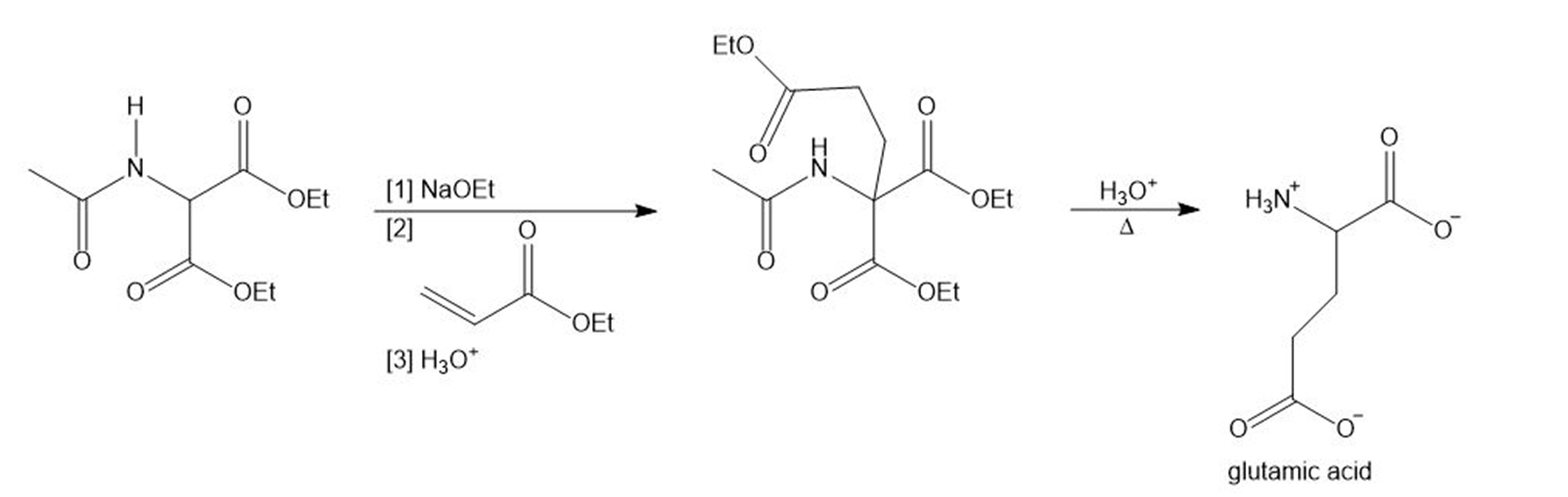
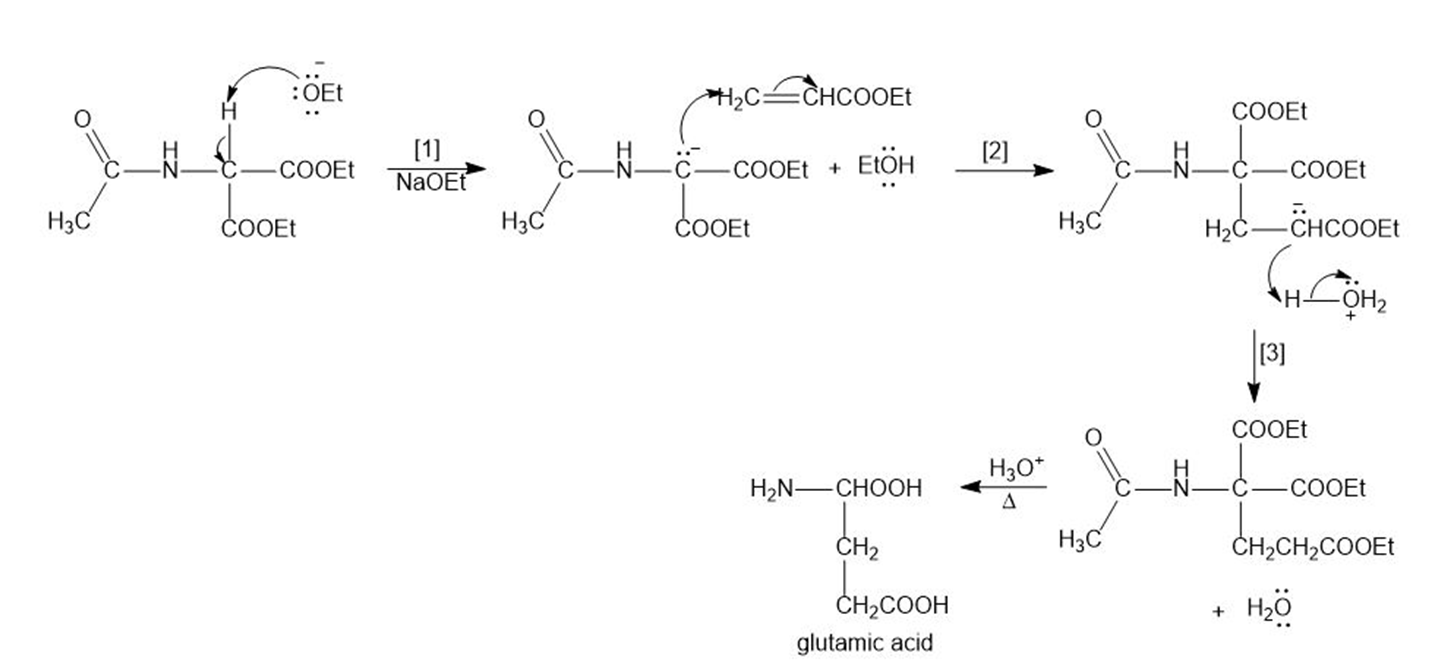
Glutamic acid is synthesized by the following reaction sequence. Draw a stepwise mechanism for Steps [1]–[3].
Short Answer
Learning Materials
Features
Discover
Chapter 29: Q44. (page 1193)
Glutamic acid is synthesized by the following reaction sequence. Draw a stepwise mechanism for Steps [1]–[3].


All the tools & learning materials you need for study success - in one app.
Get started for free
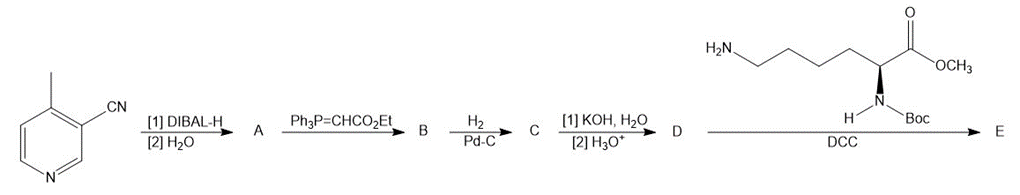
Identify A–E in the following reaction sequence.
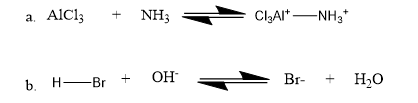
Question: Identify the nucleophile and the electrophile in the following acid–base reactions:
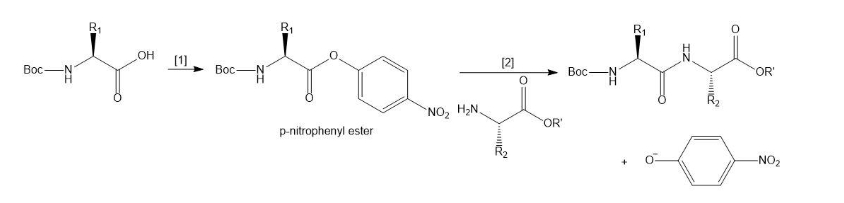
Another method to form a peptide bond involves a two-step process:
[1] Conversion of a Boc-protected amino acid to a -nitrophenyl ester.
[2] Reaction of the -nitrophenyl ester with an amino acid ester
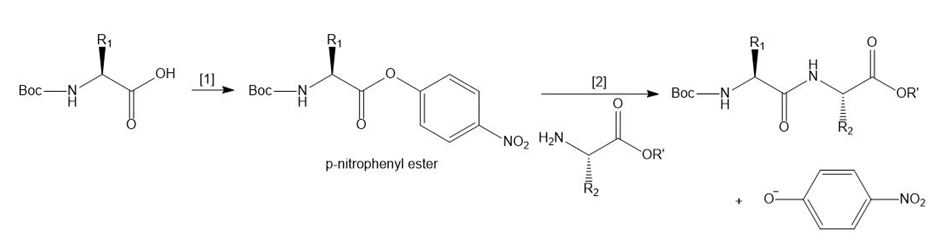
Another method to form a peptide bond involves a two-step process:
[1] Conversion of a Boc-protected amino acid to a p-nitrophenyl ester.
[2] Reaction of the p-nitrophenyl ester with an amino acid ester.
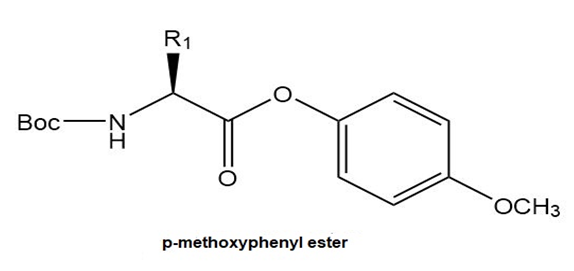
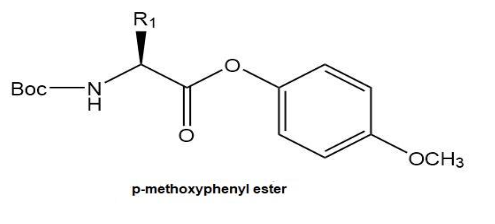
Why does a p-nitrophenyl ester “activate” the carboxy group of the first amino acid to amide formation?
Would a p-methoxyphenyl ester perform the same function? Why or why not?
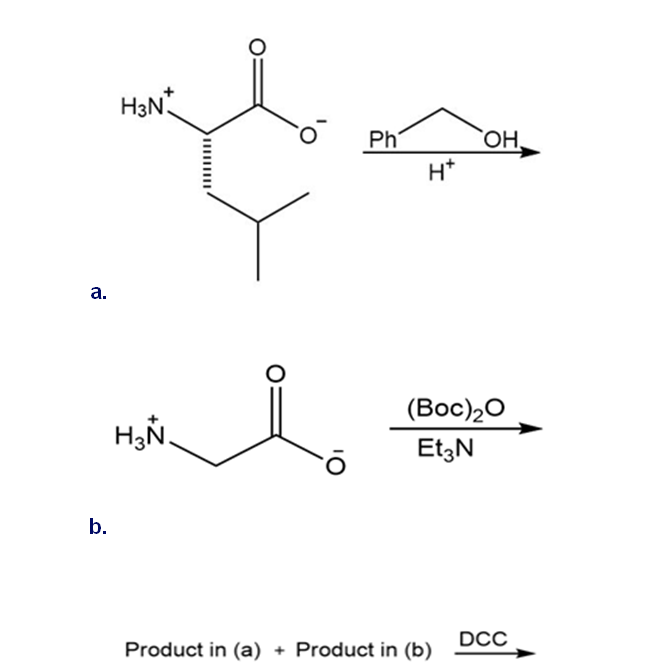
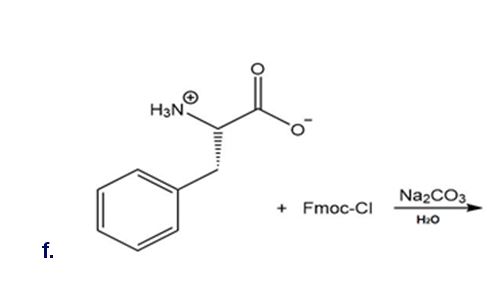
Draw the organic products formed in each reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.