Chapter 29: Q36. (page 1192)
What is the predominant form of each of the following amino acids at pH=1? What is the overall charge on the amino acid at this pH?(a)threonine ; (b)methionine; (c)aspartic acid; (d)arginine
Short Answer
(a)
(b)
(c)
(d)
Learning Materials
Features
Discover
Chapter 29: Q36. (page 1192)
What is the predominant form of each of the following amino acids at pH=1? What is the overall charge on the amino acid at this pH?(a)threonine ; (b)methionine; (c)aspartic acid; (d)arginine
(a)
(b)
(c)
(d)
All the tools & learning materials you need for study success - in one app.
Get started for free
What types of stabilizing interactions exist between each of the following pairs of amino acids?
a. Ser and Tyr
b. Val and Leu
c. two Phe residues
Besides asymmetric hydrogenation (Section 29.4), several other methods are now available for the synthesis of optically active amino acids. How might a reaction like the Strecker synthesis be adapted to the preparation of chiral amino acids?
What amino acid is formed whenis treated with the following series of reagents:
role="math" localid="1648630585393"
Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:
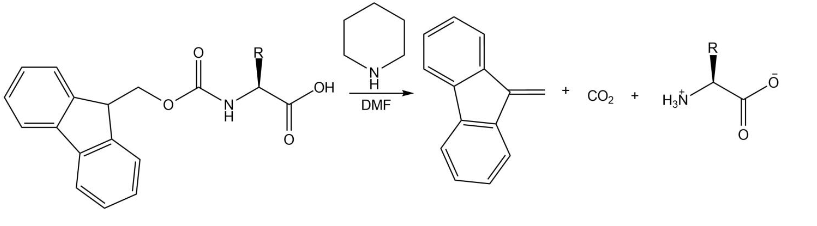
Write out the steps for the synthesis of each peptide using the Merrifield method:
What do you think about this solution?
We value your feedback to improve our textbook solutions.