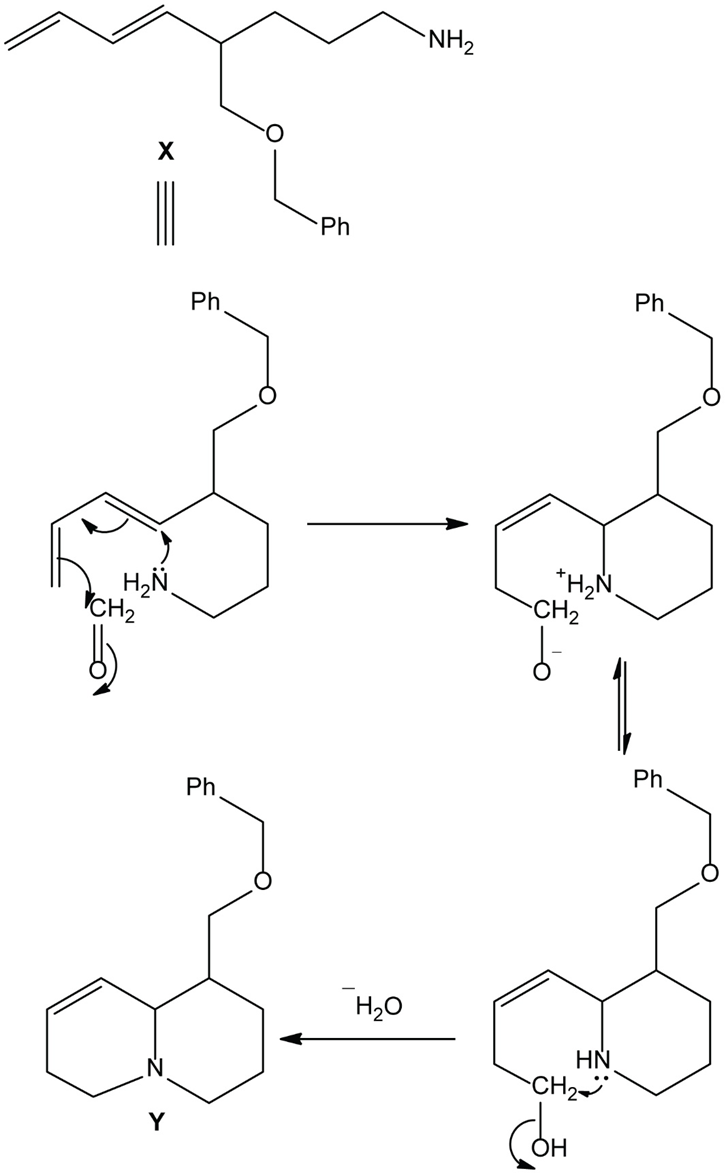
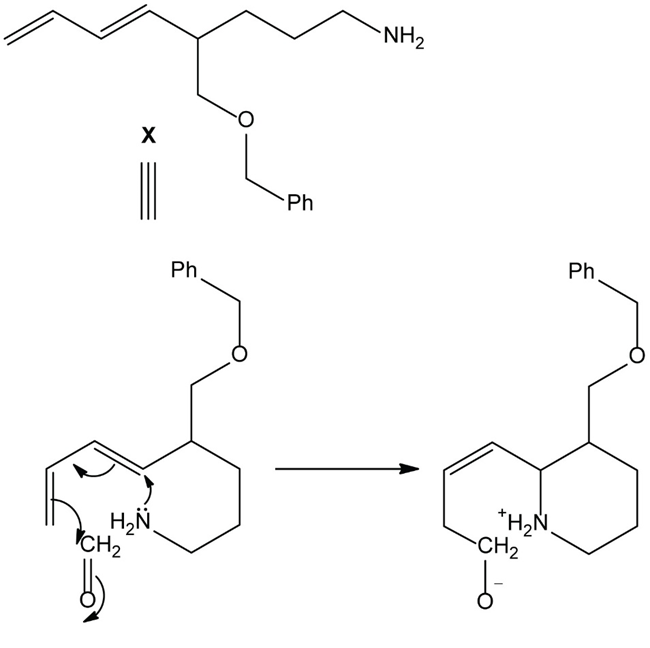
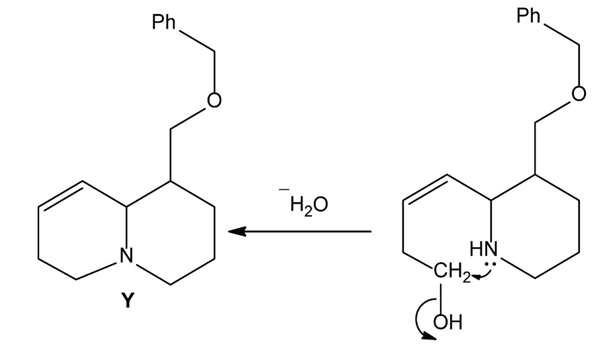
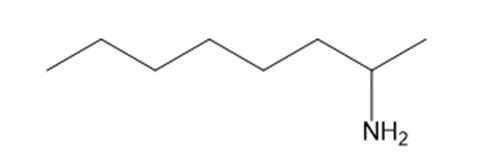
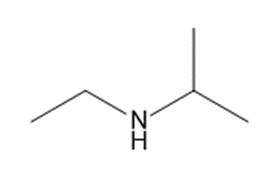
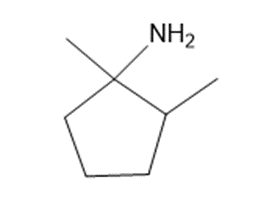
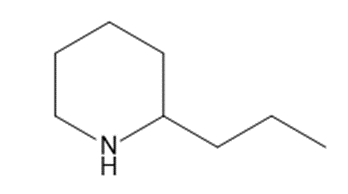
Chapter 25: 78 (page 1048)
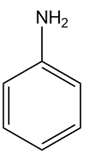
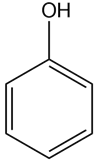
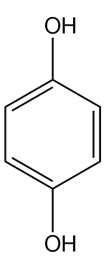
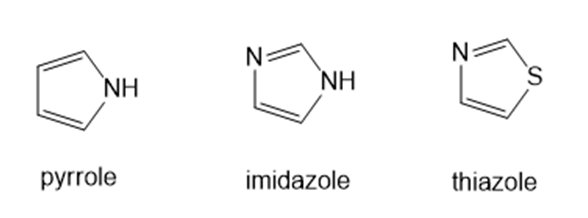
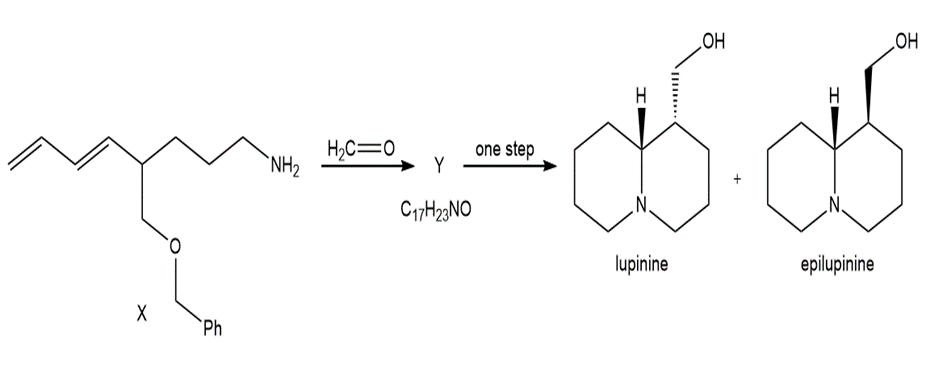
Heating compound X with aqueous formaldehyde forms Y , which has been converted to a mixture of lupinine and epilupinine, alkaloids isolated from lupin, a perennial ornamental plant commonly seen on the roadside in parts of Alaska (Section 25.10). Identify Y and explain how it is formed.
Short Answer