Chapter 25: 53 (page 1043)
Draw the products formed when each amine is treated with [1] (excess); [2] ; [3] . Indicate the major product when a mixture results.
a
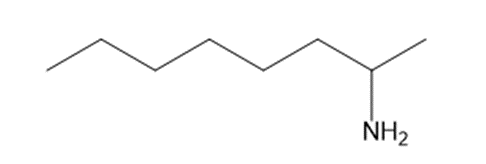
b
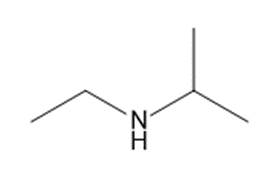
c
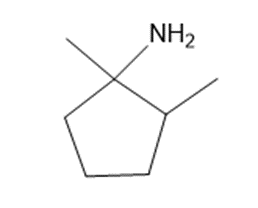
d
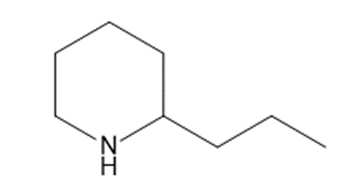
Short Answer
a
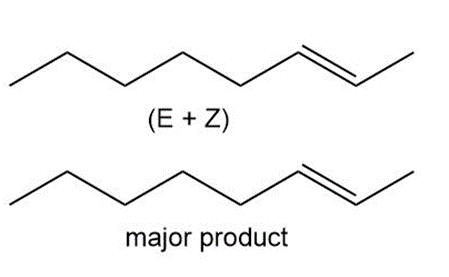
b
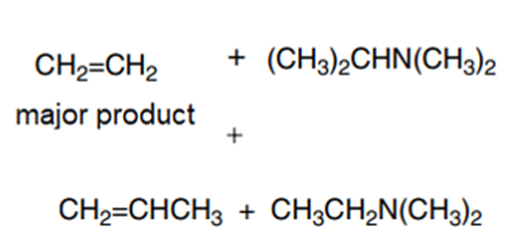
c
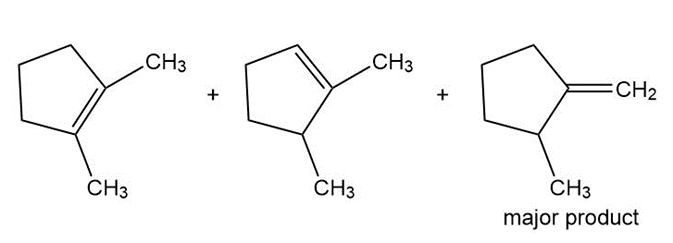
d
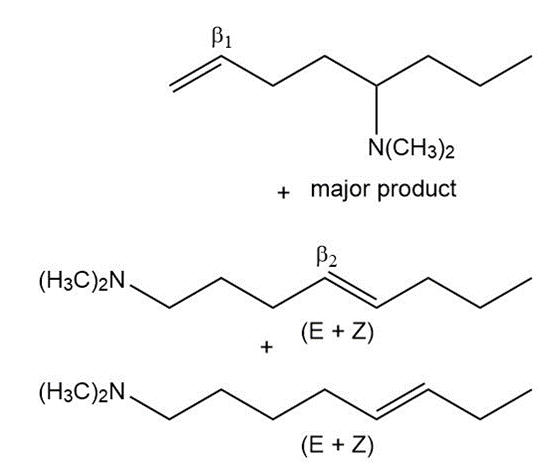
Learning Materials
Features
Discover
Chapter 25: 53 (page 1043)
Draw the products formed when each amine is treated with [1] (excess); [2] ; [3] . Indicate the major product when a mixture results.
a

b

c

d

a

b
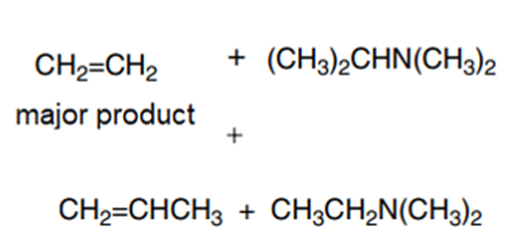
c

d

All the tools & learning materials you need for study success - in one app.
Get started for free
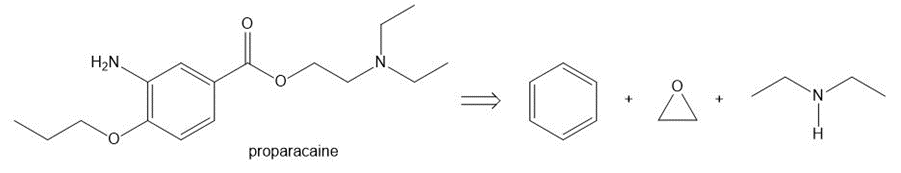
Devise a synthesis of each compound from the given starting material(s). Albuterol is a bronchodilator and proparacaine is a local anesthetic.
(a)
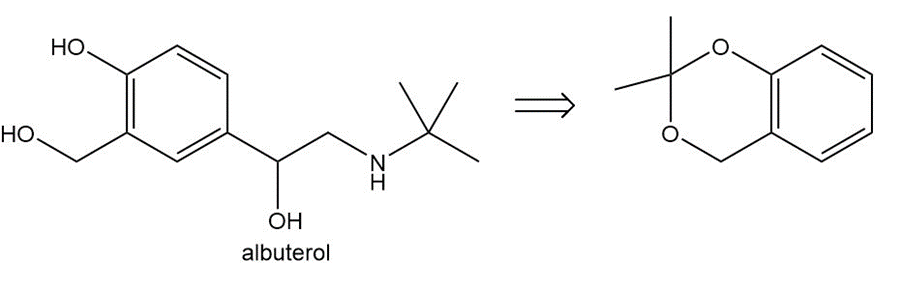
(b)
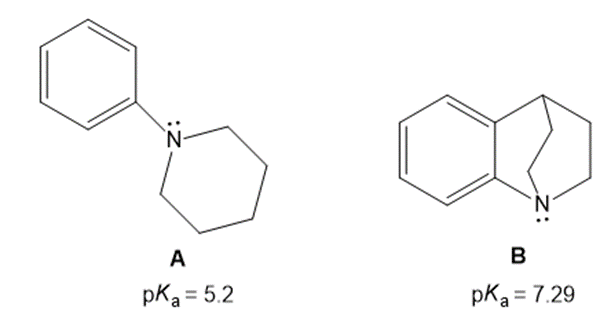
Explain the observed difference in the values of the conjugate acids of amines A and B.
Tertiary ( ) aromatic amines react with and HCl to afford products of electrophilic aromatic substitution. Draw a stepwise mechanism for this nitrosation reaction and explain why it occurs only on benzene rings with strong ortho, para activating groups.
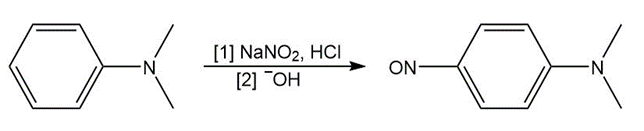
Question: Draw a structure corresponding to each name.
a. 2,4-dimethylhexan-3-amine
b. N-methylpentan-1-amine
c. N-isopropyl-p-nitroaniline
d. N-methylpiperidine
e. N,N-dimethylethanaminef. 2-aminocyclohexanoneg. N-methylanilineh. m-ethylaniline
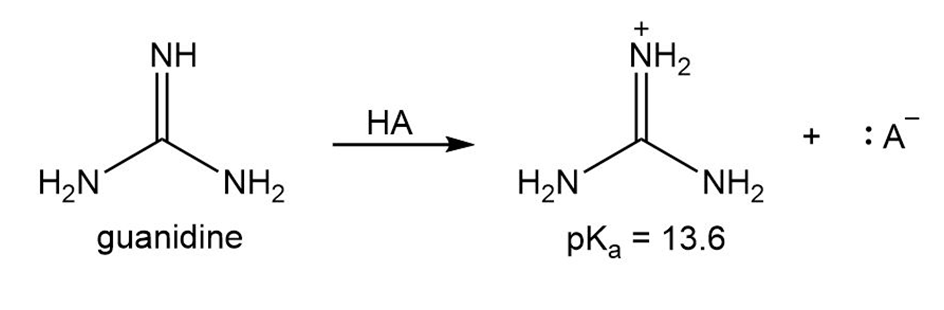
Question:The pKa of the conjugate acid of guanidine is 13.6, making it one of the strongest neutral organic bases. Offer an explanation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.