Chapter 25: 41 (page 1042)
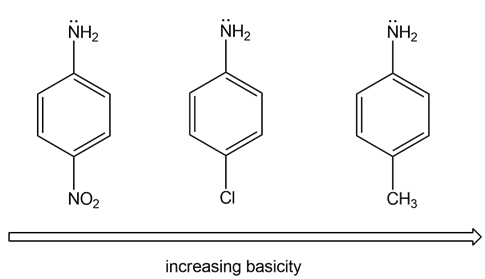
Rank the compounds in each group in order of increasing basicity.
(a)
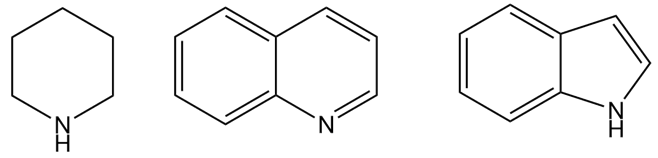
(b)

Short Answer
The order of increasing basicity
(a)
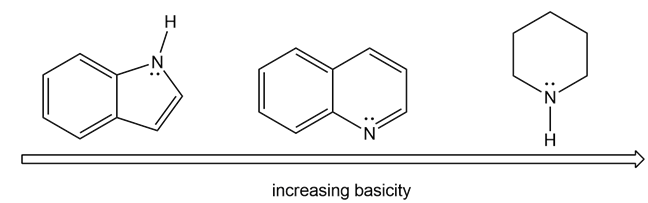
(b)
Learning Materials
Features
Discover
Chapter 25: 41 (page 1042)
Rank the compounds in each group in order of increasing basicity.
(a)

(b)

The order of increasing basicity
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for each reaction.
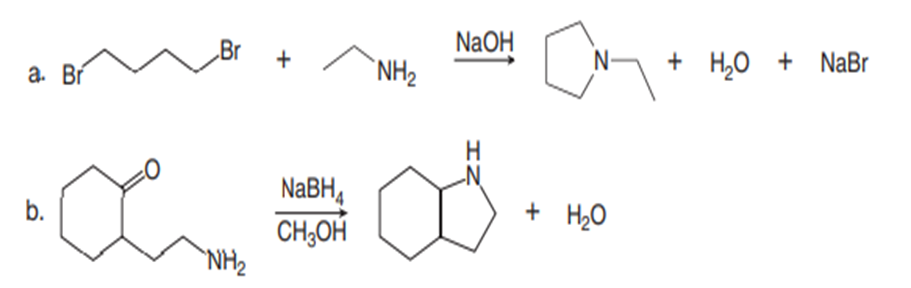
Question: Draw the products formed when each carbonyl compound reacts with the following amines:
1. CH3CH2CH2NH2
2.(CH3CH2)2NH
a.
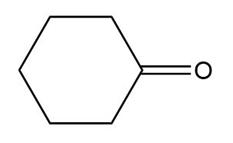
b.
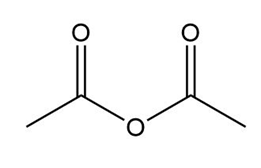
c.
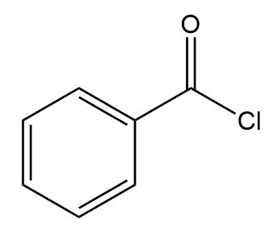
Tertiary ( ) aromatic amines react with and HCl to afford products of electrophilic aromatic substitution. Draw a stepwise mechanism for this nitrosation reaction and explain why it occurs only on benzene rings with strong ortho, para activating groups.
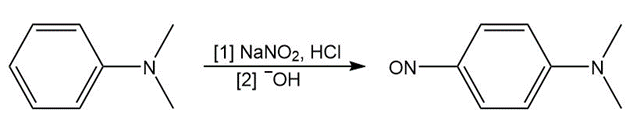
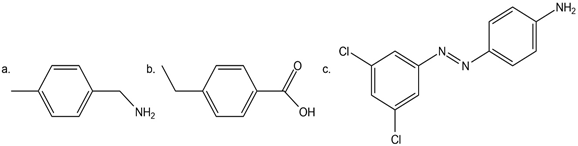
Question: Which compound in each pair is more basic:
a.
b. ?
Question: Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
What do you think about this solution?
We value your feedback to improve our textbook solutions.