Chapter 11: Q60. (page 453)
Question: Draw a stepwise mechanism for the following reaction.
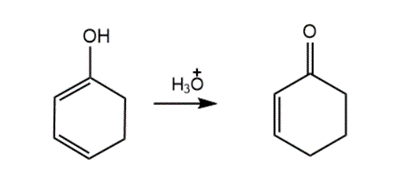
Short Answer
Answer
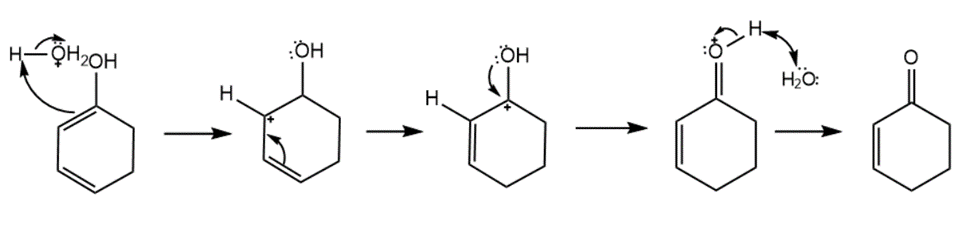
Mechanism for the given reaction
Learning Materials
Features
Discover
Chapter 11: Q60. (page 453)
Question: Draw a stepwise mechanism for the following reaction.

Answer

Mechanism for the given reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Answer the following questions about erlotinib and phomallenic acid C. Erlotinib, sold under the trade name Tarceva, was introduced in 2004 for the treatment of lung cancer. Phomallenic acid C is an inhibitor of bacterial fatty acid synthesis.

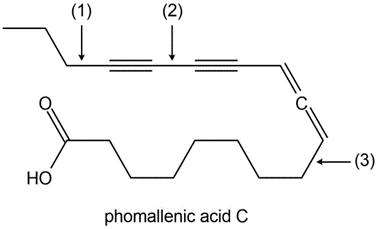
a. Which C-H bond in erlotinib is most acidic?
b.What orbitals are used to form the shortest C-C single bond in erlotinib?
c. Which H atom in phomallenic acid C is most acidic?
d. How many sp hybridized carbons are contained in phomallenic acid C?
e. Rank the labelled bonds in phomallenic acid C in order of increasing bonds strength.
Question: Draw the enol tautomer of (a) and the keto tautomer of (b).
a.
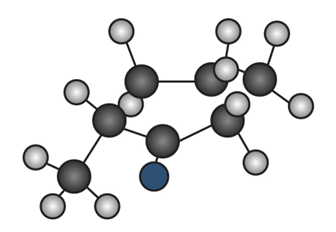
b. 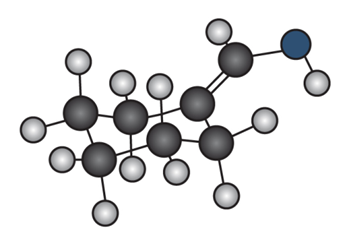
Which bases can deprotonate acetylene? The pKa values of the conjugate acids are given in parentheses.
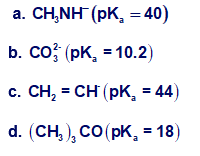
Question: Ignoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.
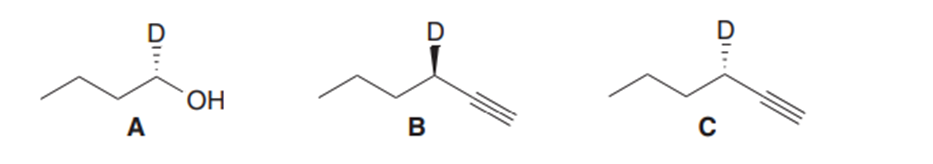
Question: What reactions are needed to convert alcohol A into either alkyne B or alkyne C?
What do you think about this solution?
We value your feedback to improve our textbook solutions.