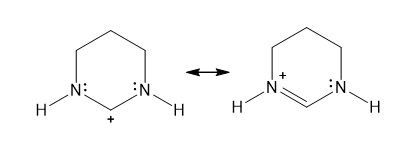
Chapter 11: 8P (page 426)
Draw additional resonance structures for each cation
(a)
(b)
(c)
Short Answer
(a)
(b)
(c)
Learning Materials
Features
Discover
Chapter 11: 8P (page 426)
Draw additional resonance structures for each cation
(a)
(b)
(c)
(a)
(b)
(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The alkyl halides must be methyl or
a.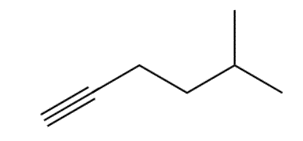
b.
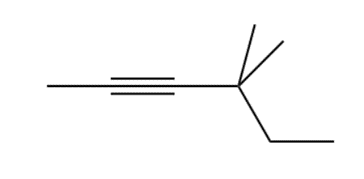
c.
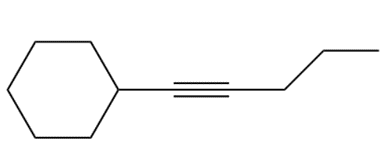
Question: Devise a synthesis of each compound. You may use HC≡CH , ethylene oxide, and alkyl halides as organic starting materials and any inorganic reagents.
a.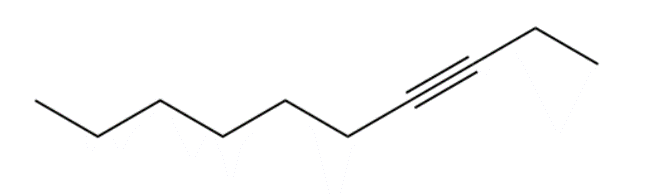
b.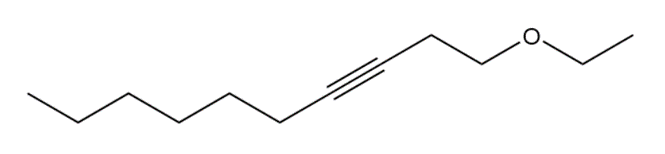
Question: Draw the enol form of each keto tautomer in parts (a) and (b), and the keto form of each enol tautomer in parts (c) and (d).
a. 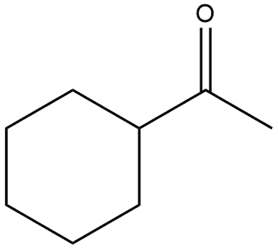
b.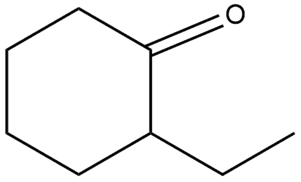
c. 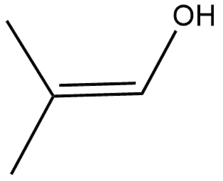
d. 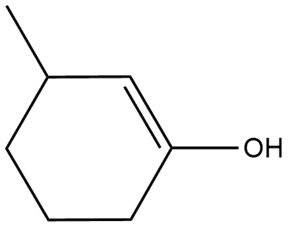
Question: Draw the products formed when hex-1-yne is treated with each reagent.

Question: Explain why 2,2,5,5-tetramethylhex-3-yne can’t be made using acetylide anions
What do you think about this solution?
We value your feedback to improve our textbook solutions.