Chapter 8: Q.37 (page 327)
What is the major stereoisomer formed when each alkyl halide is treated with ?
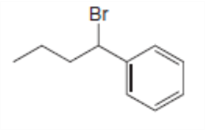
a.
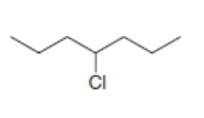
b.
Short Answer
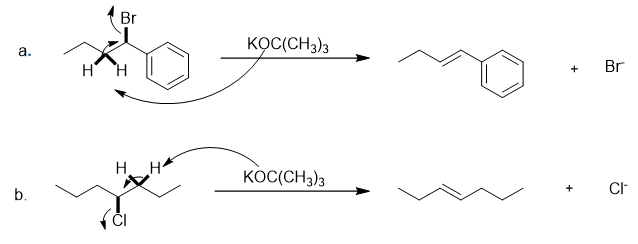
The mechanical approach of the mentioned E2 elimination is shown below:
Learning Materials
Features
Discover
Chapter 8: Q.37 (page 327)
What is the major stereoisomer formed when each alkyl halide is treated with ?

a.

b.
The mechanical approach of the mentioned E2 elimination is shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products formed when each dihalide is treated with excess .
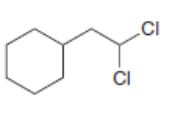
a.
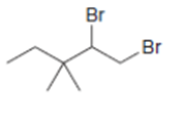
b.
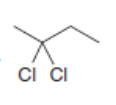
c.
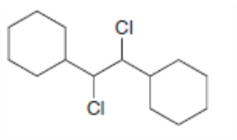
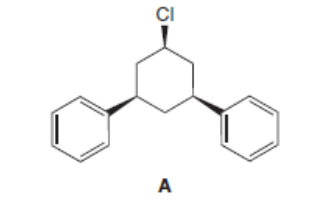
d.
Explain why compound A does not undergo an E2 elimination with a strong base.
Draw a stepwise, detailed mechanism for each reaction.
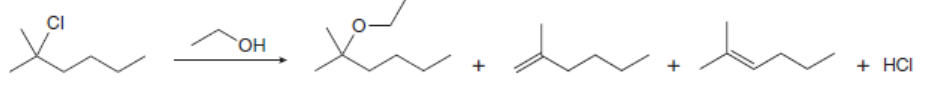
a.
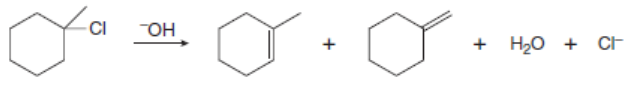
b.
For which double bonds are stereoisomers possible?
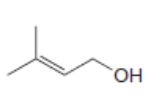
a.
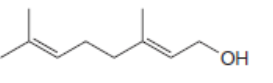
b.
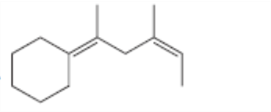
c.
Which double bonds in the following natural products can exhibit stereoisomerism? Nerolidol is isolated from the angel’s trumpet plant, caryophyllene is present in hemp, and humulene comes from hops.
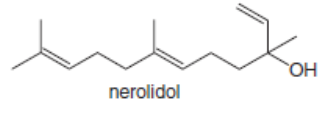
a.
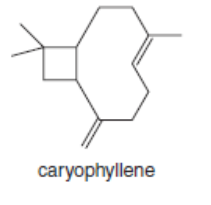
b.
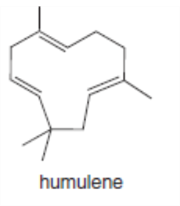
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.