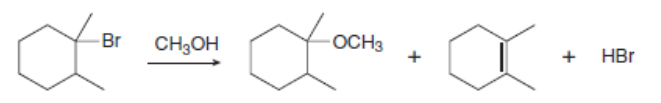
Chapter 8: Q.23 (page 323)
Draw a stepwise mechanism for the following reaction.
Short Answer
The mechanism shown for the reaction are given below:-
Learning Materials
Features
Discover
Chapter 8: Q.23 (page 323)
Draw a stepwise mechanism for the following reaction.

The mechanism shown for the reaction are given below:-

All the tools & learning materials you need for study success - in one app.
Get started for free
Label each pair of alkenes as constitutional isomers, stereoisomers, or identical.

a.

b.

c.

d.
Does cis-or trans-1-bromo-4-tert-butylcylohexane react faster in an E2 reaction?
Under certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new bonds. Product A has two sp hybridized carbon atoms, product B has one sphybridized carbon atom, and product C has none. What are the structures of A, B, and C?
Pick the reactant or solvent in each part that gives the faster elimination reaction.
a. reaction of with 1-chloro-1-methylcyclohexane or 1-chloro-3-methylcyclohexane
b. reaction of with or
c. reaction of with in or DMSO
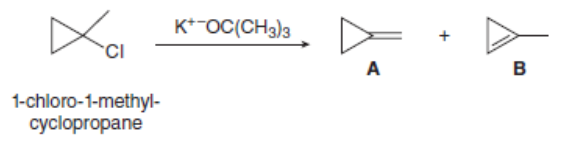
Dehydrohalogenation of 1-chloro-1-methyl cyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.
What do you think about this solution?
We value your feedback to improve our textbook solutions.