a.3-Methylhexane
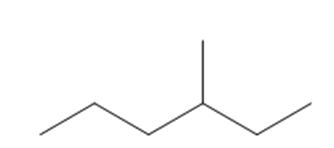
Structure of 3-methylhexane
The longest chain in the given IUPAC name is hexane, which means the compound has a six carbon-containing straight chain. The substituent methyl group is bonded at the third carbon atom of the parent chain.
b. 3,3-Dimethylpentane
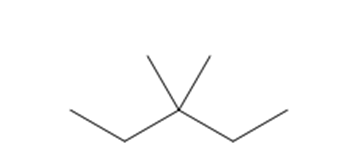
Structure of3,3-dimethylpentane
The longest chain in the given IUPAC name is pentane, which means the compound has a five carbon-containing straight chain. Two methyl groups are bonded at the third carbon atom of the parent chain.
c. 3,5,5-Trimethyloctane
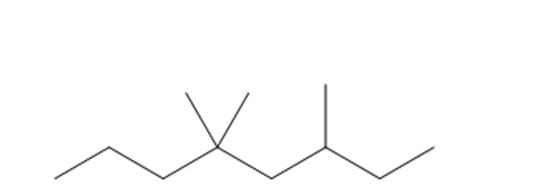
Structure of 3,5,5-trimethyloctane
The longest chain in the given IUPAC name is octane, which means the compound has an eight carbon-containing straight chain. The substituents as three methyl groups are bonded at the parent chain's third and fifth carbon atoms.
d. 3-ethyl-4-methylhexane
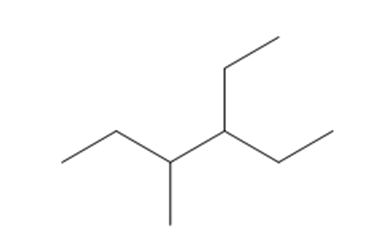
Structure of 3-ethyl-4-methylhexane
The longest chain in the given IUPAC name is hexane, which means the compound has a six carbon-containing straight chains. Ethyl and methyl groups are bonded at the parent chain's third and fourth carbon atoms, respectively.
e. 3-ethyl-5-isobutylnonane
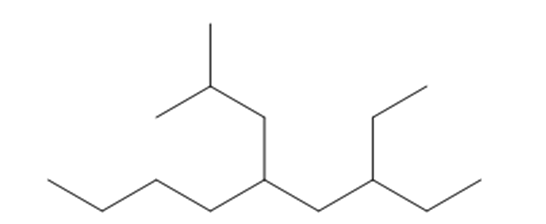
Structure of 3-ethyl-5-isobutylnonane
The longest chain in the given IUPAC name is nonane, which means the compound is a six carbon-containing straight chain. The substituents ethyl and isobutyl groups are bonded at the parent chain's third and fifth carbon atoms, respectively.

















